Abstract
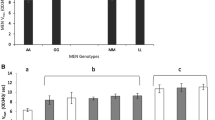
Acid phosphatase, a dimeric enzyme, in Drosophila malerkotliana was studied in isogenic flies to explore the molecular basis of heterosis. As the enzyme activity in heterozygotes is 34% more than that in the better parent (S/S), heterosis is indicated. V max, K m , and K i values are 14.60, 3.6×10−4 m, and 0.45×10−4 m, respectively, for the enzyme from F/S flies and 11.80, 4.0×10−4 m, and 0.37×10−4 m, respectively, for the enzyme from S/S flies. Thus heterosis for enzyme activity results from a better enzyme in F/S flies. The higher efficiency and better quality of the enzyme in F/S flies were traced to the heterodimeric allozyme, present only in heterozygotes. Enzyme activity, V max, K m , and K i values are 0.739, 42.1; 3.6×10−4 m, and 0.50×10−4 m, respectively, for the heterodimeric and 0.513, 36.8; 4.1×10−4 m, and 0.37×10−4 m, respectively, for the better parental homodimeric allozyme. On an equimolar basis the enzyme activity of the heterodimer is 44% higher than that of the better homodimer. The better performance of the heterodimer is probably a reflection of superior conformation resulting from interaction between component subunits (F and S polypeptides).
Similar content being viewed by others
References
Alahiotis, S. N. (1982). Adaptation of Drosophila enzymes to temperature. IV. Natural selection at the alcohol dehydrogenase locus. Genetica 5981.
Beckman, G. (1970). Placental alkaline phosphatase, relation between phenotype and enzyme activity. Hum. Hered. 2074.
Berger, E. M. (1974). Esterases of Drosophila. II. Biochemical studies of esterases-5 in D. pseudoobscura. Genetics 781157.
Catcheside, D. G., and Overton, A. (1958). Complementation between alleles in heterokaryons. Cold Spring Harbor Symp. Quant. Biol. 23137.
Chourey, P. S., and Nelson, O. F. (1979). Intra-allelic complementation at Sh locus in maize at enzyme level. Genetics 91317.
Crick, F. H., and Orgel, L. E. (1964). The theory of interallelic complementation. J. Mol. Biol. 8161.
Day, T. H., Hillar, P. C., and Clarke, B. (1974). Properties of genetically polymorphic isozymes of alcohol dehydrogenase in Drosophila melanogaster. Biochem. Genet. 11141.
Dixon, M. (1953). The determination of enzyme inhibitor constants. Biochem. J. 55170.
Ebitani, N. (1974). The variability and esterase activities of electrophoretic variants of Drosophila virilis. Dros. Info. Serv. 5131.
Gahne, B., Bengtsson, S., and Sandberg, K. (1970). Genetic control of cholin esterase activity in horse serum. Anim. Blood Groups Biochem. Genet. 1207.
Jackson, D. A., and Yanofsky (1969). The conformation and properties of dimers of tryptophan synthetase α-subunit of Escherichia coli. J. Biol. Chem. 2444526.
Koehn, R. K. (1969). Esterase heterogeneity: Dynamics of a polymorphism. Science 163943.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193265.
MacIntyre, R. J. (1971). A method of measuring activities of acid phosphatases separated by polyacrylamide gel electrophoresis. Biochem. Genet. 545.
Miller, S., Pearcy, R. W., and Berger, E. (1975). Polymorphism at the α-glycerophosphate dehydrogenase locus in Drosophila melanogaster. Properties of adult allozymes. Biochem. Genet. 13175.
Ott, L. A., and Scandalios, J. G. (1976). Genetically defined peptidases of maize. I. Biochemical characterization of allelic and non-allelic forms. Biochem. Genet. 14619.
Parin, D. (1963). Complementation between products of the β-galactosidase structural gene of Escherichia coli. Cold Spring Harbor Symp. Quant. Biol. 28529.
Parkash, R., and Gill, K. S. (1980). Biochemical characterisation of allelic acid phosphatases in Drosophila malerkotliana. Ind. J. Exp. Biol. 1861.
Scandalios, J. G., Liu, E., and Campeau, M. A. (1972). The effects of intragenic and intergenic complementation on catalase structure and function in maize. A molecular approach to heterosis. Arch. Biochem. Biophys. 153695.
Schlesinger, M., and Levinthal, C. (1963). Hybrid protein formation of E. coli alkaline phosphatase leading to in vitro complementation. J. Mol. Biol. 71.
Schwartz, D., and Laughmer, W. J. (1969). A molecular basis of heterosis. Science 166626.
Shafer, M. P., Hannon, W. H., and Levin, A. P. (1974). In vivo and in vitro complementation between Gua B and in vivo complementation between Gua A auxotrophs of Salmonella typhimurium. J. Bacteriol. 1171270.
Sumiko, N. (1973). Esterase isozymes of Drosophila virilis: Purification and properties of α- and β-esterase isozymes. Jap. J. Genet. 48119.
Trehan, K. S. (1983). Molecular Basis of Heterosis and Homeostatis Ph.D. thesis, Punjab Agricultural University, Ludhiana.
Trehan, K. S., and Gill, K. S. (1983). Sub-unit interaction, a molecular basis of heterosis. XV Int. Cong. Genet., New Delhi, Abstr., Part I, p. 391.
Trehan, K. S., and Gill, K. S. (1985). Isolation and partial purification of allozymes of acid phosphatase from heterozygotes of Drosophila malerkotliana. Dros. Info. Serv. 61174.
Trehan, K. S., and Gill, K. S. (1986). Effect of inbreeding on wing morphology in Drosophila malerkotliana. Dros. Info. Serv. 63130.
Vigue, C. L., and Johnson, F. M. (1973). Isozyme variability in species of genus Drosophila. IV. Frequency property environment relationship of allelic alcohol dehydrogeanse in D. melanogaster. Biochem. Genet. 9213.
Wills, C. J., and Nichols, L. (1971). Single gene heterosis in Drosophila revealed by inbreeding. Nature 233123.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Trehan, K.S., Gill, K.S. Subunit interaction: A molecular basis of heterosis. Biochem Genet 25, 855–862 (1987). https://doi.org/10.1007/BF00502605
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00502605