Summary
The value of 3-methoxytyramine (3-MT) as an indicator of impulse-related dopamine (DA) release has been assessed in rat corpus striatum. Moreover, the turnover of 3-MT was estimated by measuring its disappearance rate after COMT inhibition. Quantitation of 3-MT and DA was performed by gas chromatography/mass spectrometry (selected ion monitoring).
Haloperidol in doses between 0.05 and 3 mg/kg p.o. did not increase endogenous 3-MT levels at any time up to 24 h after its administration, whereas it dose-dependently increased homovanillic acid and 3,4-dihydroxyphenylacetic acid. However, in doses above 0.1 mg/kg p.o., it enhanced the accumulation of 3-MT in clorgyline-pretreated animals. Conversely, baclofen in doses of 2 mg/kg i.p. and above decreased endogeneous 3-MT levels, but reduced the accumulation of this amine only poorly at 20 mg/kg i.p. in clorgyline-pretreated rats. A number of dopamine agonists, apomorphine (0.5 mg/kg i.v.), dipropyl-ADTN (0.03 mg/kg i.v.), but not bromocriptine (1 mg/kg i.v.) reduced endogenous 3-MT levels 10 min after administration by approximately 50%.
The DA releasing agents d-amphetamine and methylphenidate showed different effects: the former increased endogenous 3-MT greatly, whereas the latter was without effect. The difference is likely to be related to the MAO inhibitory properties of amphetamine.
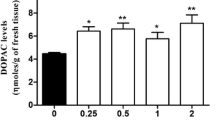
3-MT disappeared rapidly after COMT inhibition with 50 mg/kg i.v. tropolone (half-life of the initial disappearance about 1 min). The curve flattened off after 5–10 min. Turnover was calculated to be about 7 nmol/g/h, which corresponds to about a third of the turnover of DA.
Our results suggest that an important part of DA metabolism occurs through 3-MT and that this amine is very effectively deaminated by MAO-A, so effectively indeed that increased formation does not increase its endogeneous levels. It appears, therefore, that 3-MT is not suitable as an indicator of increased DA release. However, it seems to have some value for an assessment of lowered DA release.
Similar content being viewed by others
References
Bernard P, Edwards SJ, Fielding S, Robson RD, Saelens JK, Simke JP, Welch J (1975) Baclofen: an unusual CNS-active agent. Pharmacologist 17:256
Besson M-J, Cheramy A, Feltz P, Glowinski J (1971) Dopamine: spontaneous and drug-induced release from the caudate nucleus in the cat. Brain Res 32:407–424
Blum W, Richter WJ (1977) Parallel flame ionization detection —total ion current recording in capillary gas chromatography —chemical ionization mass spectrometry. J Chromatogr 132:249–259
Braestrup C (1977) Biochemical differentiation of amphetamine vs. methylphenidate and nomifensine in rats. J Pharm Pharmacol 29:463–470
Burgen ASV, Iversen LL (1965) The inhibition of noradrenaline uptake by sympathomimetic amines in the rat isolated heart. Br J Pharmacol 25:34–49
Carlsson A, Lindquist M (1963) Effect of chlorpromazine or haloperidol on formation of 3-methoxytyramine and normetanephrine in mouse brain. Acta Pharmacol Toxicol 20:140–144
Carlsson A, Lindquist M, Kehr W (1974) Postmortal accumulation of 3-methoxy-tyramine in brain. Naunyn-Schmiedeberg's Arch Pharmacol 284:365–372
Clarke DE (1980) Amphetamine and monoamine oxidase inhibition: an old idea gains new acceptance. Trends Pharmacol Sci 1:312–313
Costa E, Neff NH (1970) Isotopic and nonisotopic measurements of the rate of catecholamine biosynthesis. In: Costa E, Côté LJ, Yahr WD (eds) Biochemistry and pharmacology of the basal ganglia. Raven Press, New York, pp 141
Costa E, Neff NH (1970) Estimation of turnover rates of study the metabolic regulation of the steady state level of neuronal monoamines. In: Lajtha A (ed) Handbook of neurochemistry, vol 4. Plenum Press, New York London, pp 45
Di Giulio AM, Groppetti A, Cattabeni F, Galli CL, Maggi A, Algeri S, Ponzio F (1978) Significance of dopamine metabolites in the evaluation of drugs acting on dopaminergic neurons. Eur J Pharmacol 52:201–207
Farnebo LO, Hamberger B (1971) Drug-induced changes in the release of 3H-monoamines from field-stimulated rat brain slices. Acta Physiol Scand Suppl 371;35–44
Galli CL, Cattabeni F, Eros T, Spano PF, Algeri S, Di Giulio A, Groppetti A (1976) A mass fragmentographic assay of 3-methoxytyramine in rat brain. J Neurochem 27:795–798
Green AL, El Hait MAS (1978) Inhibition of mouse brain monoamine oxidase by (+)-amphetamine in vivo. J Pharmacol 30:262–263
Groppetti A, Parenti M, Galli CL, Bugatti A, Cattabeni F, Di Giulio AM, Racagni G (1978) 3-methoxytyramine and different neuroleptics: dissociation from HVA and DOPAC. Life Sci 23: 1763–1768
Handforth A, Sourkes TL (1975) Inhibition by dopamine agonists of dopamine accumulation following p-hydroxybutyrate treatment. Eur J Pharmacol 34:311–319
Johnston JP (1968) Some observations upon a new inhibitor of monoamine oxidase in brain tissue. Biochem Pharmacol 17:1285–1297
Kehr W (1976) 3-Methoxytyramine as an indicator of impulse-induced dopamine release in rat brain in vivo. Naunyn-Schmiedeberg's Arch Pharmacol 293:209–215
Kehr W, Speckenbach W, Zimmermann R (1977) Interaction of haloperidol and γ-butyrolactone with (+)-amphetamine-induced changes in monamine synthesis and metabolism in rat brain. J Neural Transm 40:129–147
Koe BK (1975) Monoamine oxidase inhibitors antagonize the aceleration of brain dopamine synthesis induced by neuroleptic drugs in vivo: implications for the treatment of tardive dyskinesia. Experientia 31:669–671
Lauber J, Blum W, Waldmeier PC, Richter WJ (1981) Quantitation of 3-methoxytyramine and dopamine in brain tissue by capillary gas chromatography chemical ionization mass spectrometry. Biomed Mass Spectrom to be submitted
McKenzie GM, Szerb JC (1968) The effect of dihydroxyphenyl-alanine, pheniprazine and d-amphetamine on the in vivo release of dopamine from the caudate nucleus. J Pharmacol Exp Ther 62:302–308
McLennan A (1964) The release of acetylcholine and 3-hydroxytyramine from the caudate nucleus. J Physiol (Lond) 174: 152–161
Miller HH, Shore PA, Clarke DE (1980) In vivo monoamine oxidase inhibition by d-amphetamine. Biochem Pharmacol 29:1347–1354
Nieoullon A, Cheramy A, Glowinski J (1977) An adaption of the pushpull cannula method to study the in vivo release of 3H-dopamine synthesized from 3H-tyrosine in the cat caudate nucleus: effects of various physical and pharmacological treatments. J Neurochem 28:819–828
Parkinson D, Callingham BA (1979) Substrate and inhibitor selectivity of human heart monoamine oxidase. Biochem Pharmacol 28:1639–1643
Randrup A, Braestrup C (1977) Uptake inhibition of biogenic amines by newer antidepressant drugs: relevance to the dopamine hypothesis of depression. Psychopharmacology 53:309–314
Reimann W, Zumstein A, Jackisch R, Starke K, Hertting G (1979) Effect of extracellular dopamine on the release of dopamine in the rabbit caudate nucleus: evidence for a dopaminergic feedback inhibition. Naunyn-Schmiedeberg's Arch Pharmacol 306:53–60
Roth JA, Feor K (1978) Deamination of dopamine and its O-methylated derivative by human brain monoamine oxidase. Biochem Pharmacol 27:1606–1608
Waldmeier PC, Maître L (1976) On the relevance of preferential increases of mesolimbic versus striatal dopamine turnover for the prediction of antipsychotic activity of psychotropic drugs. J Neurochem 27:589–597
Westerink BHC (1979a) Effects of drugs on the formation of 3-methoxytyramine a dopamine metabolite, in the substantia nigra, striatum, nucleus accumbens and tuberculum olfactorium of the rat. J Pharm Pharmacol 31:94–99
Westerink BHC (1979b) Further studies on the sequence of dopamine metabolism in the rat brain. Eur J Pharmacol 56:313–322
Westerink BHC, Korf J (1976a) Regional rat brain levels of 3,4-dihydroxy-phenylacetic acid and homovanillic acid: concurrent fluorometric measurement and influence of drugs. Eur J Pharmacol 38:281–291
Westerink BHC, Korf J (1976b) Turnover of acidic dopamine metabolites in striatal and mesolimbic tissue of the rat brain. Eur J Pharmacol 37:249–255
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Waldmeier, P.C., Lauber, J., Blum, W. et al. 3-methoxytyramine: Its suitability as an indicator of synaptic dopamine release. Naunyn-Schmiedeberg's Arch. Pharmacol. 315, 219–225 (1981). https://doi.org/10.1007/BF00499838
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00499838