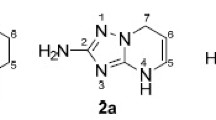
Abstract
It has been shown that 4-methylthio-, ethoxy-, and methoxyimidazotriazines and imidazotriazin-4-ones, unlike benzo-1,2,3-triazines, do not display cryptodiazonium behavior. A novel type of fission of the triazine ring to give esters and thioesters of 5-aminoimidazole-4-carboxylic acid is described.
Similar content being viewed by others
Literature Cited
V. S. Mokrushin, T. A. Pospelova, V. A. Bakulev, E. F. Golovina, S. L. Nikolaeva, and Z. V. Pushkareva, Khim. Geterotsikl. Soedin., No. 2, 247 (1984).
C. Temple, C. L. Kussner, and J. A. Montgomery, J. Org. Chem., 32, 2241 (1967).
V. S. Mokrushin, V. I. Ofitserov, T. V. Rapakova, A. G. Tsaur, and Z. V. Pushkareva, Khim. Geterotsikl. Soedin., No. 4, 556 (1976).
V. S. Mokrushin, T. A. Pospelova, and Yu. M. Shafran, Khim. Geterotsikl. Soedin., No. 11, 1548 (1983).
H. Neunhoeffer, Chem. Heterocycl. Compd., 33, 3 (1978).
H. N. E. Stevens and M. F. G. Stevens, J. Chem. Soc., C, No. 6, 765 (1970).
L. P. Kulev and V. R. Koroleva, Zh. Org. Khim., 29, 2401 (1959).
J. L. Wong and D. S. Fuchs, J. Chem. Soc., Perkin 1, No. 11, 1284 (1974).
A. Albert, J. Chem. Soc., No. 18, 2379 (1969).
B. M. Adger, S. Bradburg, M. Keating, C. M. Rees, R. S. Storr, and M. T. Williams, J. Chem. Soc., Perkin 1, No. 1, 31 (1975).
Y. F. Shealy, C. A. Krauth, and J. A. Montgomery, J. Org. Chem., 27, 2150 (1962).
Author information
Authors and Affiliations
Additional information
For Communication 14, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1248–1250, September, 1989.
Rights and permissions
About this article
Cite this article
Usova, V.K., Selezneva, I.S., Pospelova, T.A. et al. Synthesis and properties of analogs of 5(4)-aminoimidazole-4(5)-carboxamide and purines. 15. Ring opening in imidazo[4,5-d]-1,2,3-triazines. Chem Heterocycl Compd 25, 1045–1047 (1989). https://doi.org/10.1007/BF00487307
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00487307