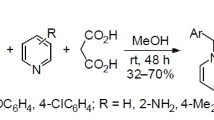
Abstract
The corresponding pyrroles and their N-vinyl derivatives were obtained by the catalyzed (by an MOH-DMSO superbase) reaction of acetylene and its crypto forms (vinyl chloride, 1,2-dichloroethane) with alkyl-2,4-, alkyl-2,5-, and alkyl-3,4-dimethylphenylketoximes. Reaction intermediates — O-vinylketoximes — were detected.
Similar content being viewed by others
Literature Cited
S. E. Korostova, S. G. Shevchenko, M. V. Sigalov, and N. I. Golovanova, Khim. Geterotsikl. Soedin., No. 4, 460 (1991).
B. A. Trofimov and A. I. Mikhaleva, N-Vinylpyrroles [in Russian], Nauka, Novosibirsk (1984).
S. E. Korostova, A. I. Mikhaleva, L. N. Sobenina, S. G. Shevchenko, R. N. Nesterenko, V. B. Modonov, and R. I. Polovnikova, Zh. Org. Khim., 24, 2538 (1988).
Author information
Authors and Affiliations
Additional information
For Communication 45 See [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1320–1323, October, 1991.
Rights and permissions
About this article
Cite this article
Aliev, I.A., Korostova, S.E., Mikhaleva, A.I. et al. Pyrroles from ketoximes and acetylene. 46. Pyrroles with sterically hindered substituents. Chem Heterocycl Compd 27, 1055–1058 (1991). https://doi.org/10.1007/BF00486796
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00486796