Abstract
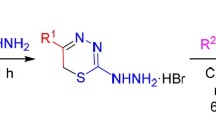
The synthesis of 3,4,4,6-tetramethyl-2-arylimino-2,3-dihydro-4H-1,3-thiazines, which have a fixed imino structure, was accomplished by two methods: by intramolecular rearrangement of 1,4,6,6-tetramethyl-3-aryl-1,2,3,6-tetrahydropyrimidine-2-thiones and by amination of 3,4,4,6-tetramethyl-2,3-dihydro-4H-1,3-thiazine-2-thione methiodide.
Similar content being viewed by others
Literature cited
P. L. Ovechkin, L. A. Ignatova, A. E. Gekhman, and B. V. Unkovskii, Khim. Geterotsikl. Soedin., 937 (1972).
B. V. Unkovskii and L. A. Ignatova, Khim. Geterotsikl. Soedin., 996 (1969).
B. V. Unkovskii, L. A. Ignatova, P. L. Ovechkin, and A. I. Vinogradova, Khim. Geterotsikl. Soedin., 1690 (1970).
J. Jansen and R. Mathes, J. Am. Chem. Soc., 77, 5431 (1955).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 941–943, July, 1972.
Rights and permissions
About this article
Cite this article
Ovechkin, P.L., Ignatova, L.A. & Unkovskii, B.V. Synthesis of substituted 2-arylimino-2,3-dihydro-4H-1,3-thiazines. Chem Heterocycl Compd 8, 856–858 (1972). https://doi.org/10.1007/BF00475218
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00475218