Abstract
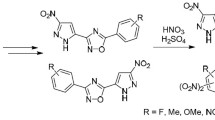
Ketones of the 3-nitro-5-R-1,2,4-triazole series react with hydrazoic acid in concentrated sulfuric acid to give triazolyl-substituted acetamides. Acid hydrolysis of the latter leads to 1-aminoalkyl-3-nitro-5-R-1,2,4-triazoles. Intramolecular cyclization with the elimination of HNO2 and the formation of 2-nitro-5,6-dihydro-1H-imidazo[2,3-b]-1,2,4-triazole was noted in the case of 1-(2′-aminoethyl)-3,5-dinitro-1,2,4-triazole.
Similar content being viewed by others
Literature cited
T. P. Kofman, T. L. Uspenskaya, N. Yu. Medvedeva, and M. S. Pevzner, Khim. Geterotsikl. Soedin., No. 7, 991 (1976).
G. I. Koldobskii, G. F. Tereshchenko, and L. I. Bagal, Zh. Org. Khim., 6, 2395 (1970).
T. P. Kofman, V. I. Manuilova, M. S. Pevzner, and T. N. Timofeeva, Khim. Geterotsikl. Soedin., No. 5, 705 (1975).
L. I. Bagal, M. S. Pevzner, and V. Ya. Samarenko, Khim. Geterotsikl. Soedin., No. 2, 269 (1970).
Author information
Authors and Affiliations
Additional information
See [1] for communication XX.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1271–1273, September, 1977.
Rights and permissions
About this article
Cite this article
Kofman, T.P., Medvedeva, N.Y., Uspenskaya, T.L. et al. Heterocyclic nitro compounds. Chem Heterocycl Compd 13, 1026–1029 (1977). https://doi.org/10.1007/BF00472466
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00472466