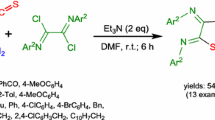
Abstract
Quaternary thiazolium salts with active methylthio groups and 2-thiazolyl and 2-pyridyl residues attached to the nitrogen atom of the thiazole ring were obtained. 2-Methyl-3-(2-pyridyl)-and 2-methyl-3-(2-thiazolyl)thiazolium salts were synthesized by replacement of the methylthio group by a methyl group. Monomethylidynecyanines and nullmethylidyne- and dimethylidynemerocyanines were obtained from salts with active methyl and methylthio groups.
Similar content being viewed by others
Literature cited
E. D. Sych and O. V. Moreiko, Khim. Geterotsikl. Soedin., 933 (1972).
L. G. S. Brooker, G. H. Keyes, R. H. Sprague, R. H. V. Dyke, E. V. Lare, G. V. Zandt, and E. L. White, J. Am. Chem. Soc., 73, 5327 (1951).
E. B. Knott, J. Chem. Soc., 1944 (1956).
Author information
Authors and Affiliations
Additional information
See [1] for communication XV.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 749–752, June, 1973.
Rights and permissions
About this article
Cite this article
Moreiko, O.V., Sych, E.D. Thiazolocyanines. Chem Heterocycl Compd 9, 687–690 (1973). https://doi.org/10.1007/BF00472307
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00472307