Abstract
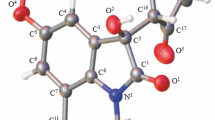
In the literature, there has appeared a communication [1] on the synthesis of 6-nitro-2, 3-dihydroquinolin-4(1H)-one (I) with mp 125–126‡C (2,4-dinitrophenylhydrazone, mp 269–270‡C) by the reaction of p-nitroaniline with acrylic acid. Quinolinone I with mp 232–234‡C (2,4-dinitrophenylhydrazone, mp 327–329‡C) was obtained by us by decarboxycyclization of N-(2-carboxy-4-nitrophenyl)-Β-alanine in acetic anhydride in the presence of potassium acetate [2].
Similar content being viewed by others
Literature cited
D. S. Chotia, S. J. Dike, A. B. Enginer, and J. R. Merchant, Indian J. Chem., 14, 323 (1976).
A. F. Bekhli and L. A. Bolotina, Khim. Geterotsikl. Soedin., No. 8, 1118 (1975).
A. Adams and D. H. Heu, J. Chem. Soc., No. 1, 255 (1949).
I. T. Braunholtz and F. G. Mann, J. Chem. Soc., Nos. 7–8, 3046 (1952).
P. I. Itteran and F. G. Mann, J. Chem. Soc., No. 1, 467 (1958).
A. F. Bekhli, L. A. Bolotina, and B. V. Lopatin, Khim. Geterotsikl. Soedin., No. 7, 954 (1978).
C. D. Hurd and S. Hayao, J. Am. Chem. Soc., 74, 5889 (1952).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1390–1392, October, 1979.
Rights and permissions
About this article
Cite this article
Bekhli, A.F., Bolotina, L.A. & Lopatin, B.V. Synthesis and constants of 6-nitro-2,3-dihydroquinolin-4(1h)-one. Chem Heterocycl Compd 15, 1120–1123 (1979). https://doi.org/10.1007/BF00471915
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00471915