Abstract
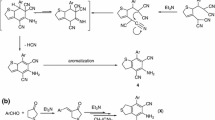
The reactions of 2, 3-dihydrothiazolo- (I) and 2, 3-dihydrothiazino-1, 3-[2, 3-b]benzthiazolium chlorides (II) with water, aqueous sodium hydroxide, aqueous alcoholic ammonia, and hydrazine hydrate are investigated. Excess alcoholic potassium hydroxide cleaves I and II to o-(Ω-mercaptoalkylamino) thiophenols, condensation of which with acetyl chloride followed by treatment of the reaction products with potassium iodide gives 2-methyl-3-(Ω-mercaptoalkyl)benzthiazolium iodides. A series of dyes is prepared from these compounds.
Similar content being viewed by others
References
E. A. Kuznetsova, S. V. Zhuravlev, and T. N. Stepanova, KhGS [Chemistry of Heterocyclic Compounds], 261, 1967.
E. A. Kuznetsova, S. V. Zhuravlev, and T. N. Stepanova, ZhOrKh, 1, 767, 1965.
W. Sexton, J. Chem. Soc., 470, 1939.
R. Reimschneider, Monatsh, 89, 683, 1958.
I. D. Kendal, Ilford. Ltd., British Patent 424559, 1935; C II, 1489, 1935.
A. I. Kiprianov and Z. N. Pazenko, ZhOKh, 19, 1523, 1949.
S. Seto and I. Ikegami, Bull. Chem. Soc. Japan, 36, 730, 1963.
C. K. Bradscher, D. F. Lohr, Jr, and W. Jones, Tetrah. Let., 22, 1723, 1963.
A. I. Kiprianov and Z. N. Pazenko, ZhOKh, 19, 1515, 1949.
L. T. Bogolyubskaya and V. A. Bogolyubskii, ZhOrKh [in press].
L. F. Deichmeister, Z. P. Sytkin, and E. B. Lifshits, ZhOKh, 22, 166, 1952.
T. G. Gnevysheva and I. I. Levkoev, DAN, 146, 1081, 1962.
F. Hamer and R. Rathbone, J. Chem. Soc., 243, 1943.
Author information
Authors and Affiliations
Additional information
For Part V see [1]
Rights and permissions
About this article
Cite this article
Kuznetsova, E.A., Bogolyubskii, V.A., Bogolyubskaya, L.T. et al. Synthesis of 2-mercaptobenzthiazole derivatives. Chem Heterocycl Compd 3, 659–661 (1967). https://doi.org/10.1007/BF00468339
Issue Date:
DOI: https://doi.org/10.1007/BF00468339