Summary
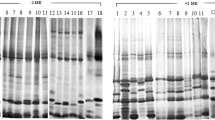
Two forms of aminopeptidase (AmP) were found, by conventional zone electrophoresis, to be present in all tissues at various stages of normal development and differentiation of Pisum sativum. One of the enzymes (AmP-1) proved to be polymorphic, with alternate electrophoretic forms existing in different inbred pea strains, while the other enzyme (AmP-2) was found to be monomorphic. The AmP-1 variants are under the control of two codominant alleles (AmP-1 F and AmP-1 S) at the AmP-1 locus. The AmP-2 enzyme is most likely controlled by a separate genetic locus. Substrate specificity studies, using various α-amino acid naphthylamides as substrates, showed that the aminopeptidases of Pisum are not specific for leucine N-terminal residues. The AmP-1 and AmP-2 enzymes behaved quite differently with respect to substrate specificity and metal ion inhibition, suggesting differences in the biological function and relatedness of the two enzymes.
Similar content being viewed by others
References
Beckman, L., G. Bjorling, and C. Christodoulou: Multiple molecular forms of leucine aminopeptidase in man. Acta genet. (Basel) 15, 218–223 (1966).
Behal, F. J., and J. D. Folds: Arylamidase of Neisseria catarrhalis. Arch. Biochem. 121, 364–371 (1967).
Goldbarg, J. A., and A. M. Rutenburg: The colorimetric determination of leucine aminopeptidase in urine and serum of normal subjects and patients with cancer and other diseases. Cancer (Philad.) 11, 283–291 (1958).
Hunter, N. W.: Effect of certain cations on activity of leucine naphthylamidases of Paramecium caudatum. Canad. J. Microbiol. 13, 1133–1138 (1967).
Johnson, F. M., and R. K. Sakai: A leucine aminopeptidase polymorphism in Drosophila buskii. Nature (Lond.) 203, 373–374 (1964).
Lowry, O. H., N. J. Rosebrough, A. L. Farr, and R. J. Randall: Protein measurement with the Folin phenol reagent. J. biol. Chem. 193, 265–275 (1951).
Mottet, N. K.: Activity of aminopeptidases during early limb bud differentiation in the chick embryo. J. exp. Zool. 165, 279–292 (1967).
Nachlas, M., T. P. Goldstein, and A. M. Seligman: An evaluation of aminopeptidase specificity with seven chromogenic substrates. Arch. Biochem. 97, 223–231 (1962).
Sakai, R. K., D. A. Tung, and J. G. Scandalios: Genetic and developmental studies of aminopeptidases in Drosophila melanogaster. Genetics (Abstr.) 60, 219–220 (1968).
Scandalios, J. G.: Tissue-specific isozyme variations in maize. J. Hered. 55, 281–285 (1964).
—: Leucine aminopeptidase in maize development. J. Hered. 56, 177–180 (1965).
—: Human serum leucine aminopeptidase: Variation in pregnancy and in disease states. J. Hered. 58, 153–156 (1967).
—: The genetic control of multiple molecular forms of enzymes in plants: A review. Biochem. Genet. 3, 37–79 (1969).
Smith, E. L., and R. L. Hill: Leucine aminopeptidase. In: The enzymes, vol. 4, p. 37–62, edit. by P. D. Boyer, H. Lardy, and K. M. Myrbach. New York: Academic Press 1960.
Author information
Authors and Affiliations
Additional information
Communicated by G. Melchers
This investigation was supported by the U.S. Atomic Energy Commission under Contract No. AT(11-1)1338.
Rights and permissions
About this article
Cite this article
Scandalios, J.G., Espiritu, L.G. Mutant aminopeptidases of Pisum sativum . Molec. Gen. Genet. 105, 101–112 (1969). https://doi.org/10.1007/BF00445679
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00445679