Abstract
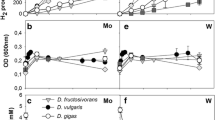
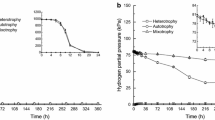
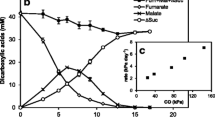
H2 production from glucose by Ruminococcus albus was almost completely inhibited by 10−5 M molybdate only when sulfide was present in the growth medium. Inhibition was accompanied by a significant increase in the production of formate. Extracts of molybdate-sulfide-grown cells did not contain hydrogenase activity. Active enzyme in extracts of uninhibited cells was not inhibited by the molybdate-sulfide-containing growth medium. The results indicate that a complex formed from molybdate and sulfide prevents the formation of active hydrogenase and electrons otherwise used to form H2 are used to reduce CO2 to formate. Growth was significantly inhibited when molybdate was increased to 10−4 M. Reversal of growth inhibition but not inhibition of H2 production occurred between 10−4 and 10−3 M molybdate. H2 production by R. bromei but not by R. flavefaciens, Butyrivibrio fibrisolvens, Veillonella alcalescens, Klebsiella pneumoniae and Escherichia coli was inhibited by molybdate and sulfide.
Similar content being viewed by others
References
Andreesen, J. R., El Ghazzawi, E., Gottschalk, G.: The effect of ferrous ions, tungstate and selenite on the level of formate dehydrogenase in Clostridium formicoaceticum and formate synthesis from CO2 during pyruvate fermentation. Arch. Microbiol. 26, 103–118 (1974)
Andreesen, J. R., Ljungdahl, L. G.: Formate dehydrogenase of Clostridium thermoaceticum: incorporation of selenium-75, and the effects of selenite, molybdate, and tungstate on the enzyme. J. Bacteriol. 116, 867–873 (1973)
Bonnichsen, R.: Ethanol. Determination with alcohol dehydrogenase and DPN. In: Methods of enzymatic analysis, (H. U. Bergmeyer, ed.), pp. 285–287. New York: Academic Press Inc. 1965
Bradford, M. M.: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976)
Chen, M., Wolin, M. J.: Influence of CH4 production by Methanobacterium ruminantium on the fermentation of glucose and lactate by Selenomonas ruminantium. Appl. Environ. Microbiol. 34, 756–759 (1977)
Glass, T. L., Bryant, M. P., Wolin, M. J.: Partial purification of ferredoxin from Ruminococcus albus and its role in pyruvate metabolism and reduction of nicotinamide adenine dinucleotide by H2. J. Bacteriol. 131, 463–472 (1977)
Leonhardt, U., Andreesen, J. R.: Some properties of formate dehydrogenase, accumulation and incorporation of 185W-tungsten into proteins of Clostridium formicoaceticum. Arch. Microbiol. 115, 277–284 (1977)
Miller, T. L.: The pathway of formation of acetate and succinate from pyruvate by Bacteroides succinogenes. Arch. Microbiol. 117, 145–152 (1978)
Miller, T. L., Wolin, M. J.: Formation of hydrogen and formate by Ruminococcus albus. J. Bacteriol. 116, 836–846 (1973)
Miller, T. L., Wolin, M. J.: A serum bottle modification of the Hungate technique for cultivating obligate anaerobes. Appl. Microbiol. 27, 985–987 (1974)
Müller, A., Glemser, O., Dieman, E., Hofmeister, H.: Bildung und Zerfall von Chalkogenoanionen der Übergangsmetalle in wässriger Lösung. Zeitschr. Anorgan. Allg. Chemie 371, 74–80 (1969)
Rabinowitz, J. C., Pricer, W. E. Jr.: An enzymatic method for the determination of formic acid. J. Biol. Chem. 229, 321–328 (1957)
Rose, I. A., Grunberg-Manago, M., Korey, S. R., Ochoa, S.: Enzymatic phosphorylation of acetate. J. Biol. Chem. 211, 737–756 (1954)
Thauer, R. K., Fuchs, G., Jungermann, K.: Role of iron-sulfur proteins in formate metabolism. In: Iron-sulfur proteins, Vol. 3 (W. Lovenberg, ed.), pp. 121–156. New York: Academic Press, Inc. 1977
Zehnder, A. J. B., Wuhrman, K.: Titanium (III) citrate as a non-toxic oxidation-reduction buffering system for the culture of obligate anaerobes. Science 214, 1165–1166 (1976)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolin, M.J., Miller, T.L. Molybdate and sulfide inhibit H2 and increase formate production from glucose by Ruminococcus albus . Arch. Microbiol. 124, 137–142 (1980). https://doi.org/10.1007/BF00427718
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00427718