Summary
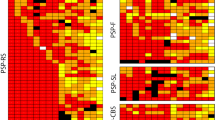
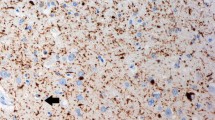
Progressive supranuclear palsy is characterized neuropathologically by the presence of high densities of neurofibrillary tangles in several subcortical structures. In some cases, neurofibrillary tangles have also been described in the cerebral cortex. We performed a quantitative regional and laminar analysis of the distribution of these lesions in six cases of progressive supranuclear palsy. We observed that the neurofibrillary tangle distribution in the cerebral cortex was largely confined to the hippocampal formation. In particular, in all the cases neurofibrillary tangles were observed in the granule cell layer of the dentate gyrus. In the prefrontal and inferior temporal cortex, neurofibrillary tangles were predominantly distributed in layers II and III. In addition, there were moderate-to-high neurofibrillary tangle densities in the primary motor cortex. This localization pattern contrasts with the neurofibrillary tangle distribution observed in the cerebral cortex of Alzheimer's disease cases, where tangles are denser in layer V than in layer III, and where the primary motor cortex and the dentate gyrus are usually not involved. These results suggest that specific elements of the cortical circuitry might be differentially vulnerable in progressive supranuclear palsy as compared to Alzheimer's disease.
Similar content being viewed by others
References
Akashi T, Arima K, Maruyama N, Ando S, Inose T (1989) Severe cerebral atrophy in progressive supranuclear palsy: a case report. Clin Neuropathol 8:195–199
Albert ML, Feldman RG, Willis AL (1974) The ‘subcortical dementia’ of progressive supranuclear palsy. J Neurol Neurosurg Psychiatry 37:121–130
Arnold SE, Hyman BT, Flory J, Damasio AR, Van Hoesen GW (1991) The topographical and neuroanatomical distribution of neurofibrillary tangles and neuritic plaques in the cerebral cortex of patients with Alzheimer's disease. Cerebral Cortex 1:103–116
Behrman S, Carroll JD, Jonata I, Matthews WB (1969) Progressive supranuclear palsy-Clinico-pathological study of four cases. Brain 92:663–678
Bouras C, Hof PR, Guntern R, Morrison JH (1990) Down's syndrome (DS), dementia pugilistica (DP), and Alzheimer's disease (AD): a quantitative neuropathologic comparison. Proc Soc Neurosci 16:1264
Bugiani O, Mancardi GL, Brusa A, Ederli A (1979) The fine structure of subcortical neurofibrillary tangles in progressive supranuclear palsy. Acta Neuropathol (Berl) 45:147–152
Constantinidis J, Tissot R, De Ajuriaguerra J (1970) Dystonie oculo-cervico-faciale ou paralysie progressive supranucléaire de Steele-Richardson-Olszewski — Pseudo-paralysie du regard, troubles visuo-spatiaux, pseudo-démence, altérations neuronales. Rev Neurol (Paris) 122:249–262
D'Antona R, Baron JC, Samson Y, Serdaru M, Viader F, Agid Y, Cambier J (1985) Subcortical dementia — Frontal cortex hypometabolism detected by positron tomography in patients with progressive supranuclear palsy. Brain 108:785–799
Défossez A, Beauvillain JC, Delacourte A, Mazzuca M (1988) Alzheimer's disease: a new evidence for common epitopes between microtubule associated protein tau and paired helical filaments (PHF): demonstration at the electron microscope by a double immunogold labelling. Virchows Arch [A] 413:141–145
Delacourte A, Flament S, Dibe EM, Hublau P, Sablonnière B, Hémon B, Scherrer V, Défossez A (1990) Pathological proteins tau 64 and 69 are specifically expressed in the somatodendritic domain of the degenerating cortical neurons during Alzheimer's disease: demonstration with a panel of antibodies against tau proteins. Acta Neuropathol 80:111–117
Dix MR, Harrison MJG, Lewis PD (1971) Progressive supranuclear palsy (the Steele-Richardson-Olszewski syndrome) —A report of 9 cases with particular reference to the mechanism of the oculomotor disorder. J Neurol Sci 13:237–256
Duyckaerts C, Hauw JJ, Bastenaire F, Piette F, Poulain C, Rainsard V, Javoy-Agid F, Berthaux P (1986) Laminar distribution of cortical senile plaques in senile dementia of the Alzheimer type. Acta Neuropathol (Berl) 70:249–256
Flament S, Delacourte A, Delaère P, Duyckaerts C, Hauw JJ (1990) Correlation between microscopical changes and tau 64 and 69 biochemical detection in senile dementia of the Alzheimer type. Tau 64 and 69 are reliable markers of the neurofibrillary degeneration. Acta Neuropathol 80:212–215
Flament S, Delacourte A, Verny M, Hauw JJ, Javoy-Agid F (1991) Abnormal tau proteins in progressive supranuclear palsy — Similarities and differences with the neurofibrillary degeneration of the Alzheimer type. Acta Neuropathol 81:591–596
Gallyas F (1971) Silver staining of Alzheimer's neurofibrillary changes by mean of physical development. Acta Morphol Acad Sci Hung 19:1–8
Ghatak NR, Nochlin D, Hadfield MG (1980) Neurofibrillary pathology in progressive supranuclear palsy. Acta Neuropathol (Berl) 52:73–76
Globus JH (1927) The Cajal and Hortega glia staining methods. A new step in the preparation of formaldehyde-fixed material. Arch Neurol Psychiatry 18:263–271
Golaz J, Bouras C, Hof PR (1991) Motor cortex involvement in presenile dementia: report of a case. J Geriatr Psychiatr Neurol 5:85–92
Guntern R, Bouras C, Hof PR, Vallet PG (1992) An improved thioflavine S method for neurofibrillary tangles and senile plaques in Alzheimer's disease. Experientia 48:8–10
Hauw JJ, Verny M, Delaère P, Cervera P, He Y, Duyckaerts C (1990) Constant neurofibrillary changes in the neocortex in progressive supranuclear palsy. Basic differences with Alzheimer's disease and aging. Neurosci Lett 119:182–186
Hirano A (1990) Amyotrophic lateral sclerosis on Guam. In pursuit of pathology over 30 years. In: Rose FC, Norris FH (eds) ALS. New advances in toxicology and epidemiology. Smith-Gordon, London, pp 103–111
Hof PR, Bouras C (1991) Object recognition deficit in Alzheimer's disease: possible disconnection of the occipitotemporal component of the visual system. Neursci Lett 122:53–56
Hof PR, Morrison JH (1990) Quantitative analysis of a vulnerable subset of pyramidal neurons in Alzheimer's disease: II. Primary and secondary visual cortex. J Comp Neurol 301:55–64
Hof PR, Bouras C, Constantinidis J, Morrison JH (1989) Balint's syndrome in Alzheimer's disease: specific disruption of the occipito-parictal visual pathway. Brain Res 493:368–375
Hof PR, Bouras C, Constantinidis J, Morrison JH (1990) Selective disconnection of specific visual association pathways in cases of Alzheimer's disease presenting with Balint's syndrome. J Neuropathol Exp Neurol 49:168–184
Hof PR, Cox K, Morrison JH (1990) Quantitative analysis of a vulnerable subset of pyramidal neurons in Alzheimer's disease: I. Superior frontal and inferior temporal cortex. J Comp Neurol 301:44–54
Hof PR, Perl DP, Steele JC, Janssen W, Morrison JH (1990) Quantitative neuropathologic analysis of ALS-PD cases from Guam. Proc Soc Neurosci 16:1264
Hof PR, Knabe R, Bovier P, Bouras C (1991) Neuropathological observations in a case of autism presenting with self injury behavior. Acta Neuropathol 82:321–326
Hof PR, Perl DP, Loerzel AJ, Morrison JH (1991) Neurofibrillary tangle distribution in the cerebral cortex of parkinsonism-dementia cases from Guam: differences with Alzheimer's disease. Brain Res 564:306–313
Ishino H (1976) Frequency of Alzheimer's neurofibrillary tangles in the cerebral cortex in progressive supranuclear palsy. Folia Psychiatr Neurol Jpn 78:463–470
Ishino H, Otsuki S (1976) Frequency of Alzheimer's neurofibrillary tangles in the cerebral cortex in progressive supranuclear palsy (subcortical argyrophilic dystrophy). J Neurol Sci 28:309–316
Ito H, Goto S, Hirano A, Yen SHC (1991) Immunohistochemical study of the hippocampus in parkinsonism-dementia complex on Guam. J Geriatr Psychiatr Neurol 4:134–142
Jellinger K (1971) Progressive supranuclear palsy (subcortical argyrophilic dystrophy). Acta Neuropathol (Berl) 19:347–352
Lewis DA, Campbell MJ, Terry RD, Morrison JH (1987) Laminar and regional distribution of neurofibrillary tangles and neuritic plaques in Alzheimer's disease: a quantitative study of visual and auditory cortices. J Neurosci 7:1799–1808
Mitsuyama Y, Seyama S (1981) Frequency of Alzheimer's neurofibrillary tangle in the brains of progressive supranuclear palsy, postencephalitic parkonsonism, Alzheimer's disease, senile demential and non-demented elderly people. Folia Psychiat Neurol Jpn 35:189–204
Morrison JH, Hof PR, Campbell MJ, De Lima AD, Voigt T, Bouras C, Cox K, Young WG (1990) Cellular pathology in Alzheimer's disease: implications of corticocortical disconnection and differential vulnerability. In: Rapoport SR, Petit H, Leys D, Christen Y (eds) Imaging, cerebral topography and Alzheimer's disease. Springer, Berlin Heidelberg New York Tokyo, pp 19–40
Mutrux S (1947) Diagnostic différentiel histologique de la maladie d'Alzheimer et de la démence sénile-Pathophobie de la zone de projection corticale. Monatsschr Psychiatr Neurol 113:100–117
Pearson RCA, Esiri MM, Hiorns RW, Wilcock GK, Powell TPS (1985) Anatomical correlates of the distribution of the pathological changes in the neocortex in Alzheimer's disease. Proc Natl Acad Sci USA 82:4531–4534
Probst A, Dufresne JJ (1975) Paralysie supranucléaire progressive (ou dystonie oculo-facio-cervicale). Schweiz Arch Neurol Neurochir Psychiatr 116:107–134
Probst A, Langui D, Lautenschlager C, Ulrich J, Brion JP, Anderton BH (1988) Progressive supranuclear palsy: extensive neuropil threads in addition to neurofibrillary tangles —Very similar antigeneicity of subcortical neuronal pathology in progressive supranuclear palsy and Alzheimer's disease. Acta Neuropathol 77:61–68
Rogers J, Morrison JH (1985) Quantitative morphology and regional and laminar distribution of senile plaques in Alzheimer's disease. J Neurosci 5:2801–2808
Sasaki S, Maruyama S, Toyoda C (1991) A case of progressive supranuclear palsy with widespread senile plaques. J Neurol 238:345–348
Schmidt ML, Lee VMY, Hurtig H, Trojanowski JQ (1988) Properties of antigenic determinants that distinguish neurofibrillary tangles in progressive supranuclear palsy and Alzheimer's disease. Lab Invest 59:460–466
Shin RW, Kitamoto T, Tateishi J (1991) Modified tau is present in younger nondemented persons: a study of subcortical nuclei in Alzheimer's disease and progressive supranuclear palsy. Acta Neuropathol 81:517–523
Steele JC (1972) Progressive supranuclear palsy. Brain 95:693–704
Steele JC, Richardson JC, Olszewski J (1964) Progressive supranuclear palsy — A heterogeneous degeneration involving brain stem, basal ganglia and cerebellum, with vertical gaze and pseudobulbar palsy, nuchal dystonia and dementia. Arch Neurol 10:333–359
Tagliavini F, Pilleri G, Bouras C, Constantinidis J (1984) The basal nucleus of Meynert in patients with progressive supranuclear palsy. Neurosci Lett 44:37–42
Tellez-Nagel I, Wisniewski HM (1973) Ultrastructure of neurofibrillary tangles in Steele-Richardson-Olszewski syndrome. Arch Neurol 29:324–327
Tomonaga M (1977) Ultrastructure of neurofibrillary tangles in progressive supranuclear palsy. Acta Neuropathol (Berl) 37:177–181
Vallet PG, Guntern R, Hof PR, Golaz J, Delacourte A, Robakis NK, Bouras C (1992) A comparative study of histological and immunohistochemical methods for neurofibrillary tangles and senile plaques in Alzheimer's disease. Acta Neuropathol 83:170–178
Van Essen DC (1985) Functional organization of the primate visual cortex. In: Peters A, Jones EG (eds) Cerebral cortex, vol 3. Plenum, New York, pp 259–329
Yagishita S, Itoh Y, Amano N, Nakano T, Saitoh A (1979) Ultrastructure of neurofibrillary tangles in progressive supranuclear palsy. Acta Neuropathol (Berl) 48:27–30
Author information
Authors and Affiliations
Additional information
Supported in part by the Brookdale Foundation and the American Health Assistance Foundation (to PRH), and ADERMA and INSERM clinical network CAR 489016 (to AD)
Rights and permissions
About this article
Cite this article
Hof, P.R., Delacourte, A. & Bouras, C. Distribution of cortical neurofibrillary tangles in progressive supranuclear palsy: A quantitative analysis of six cases. Acta Neuropathol 84, 45–51 (1992). https://doi.org/10.1007/BF00427214
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00427214