Abstract
Although muscle and nerve are reasonably well protected against active oxygen and related free radicals, environmental or inherited malfunctions can overpower their defences. Active oxygen is involved in many neuropathies and myopathies. In every case the damage is caused by agents which exert effects disproportionately greater than the quantites encountered, through a variety of amplification mechanisms. We can categorize these amplification mechanisms as follows: (a) non-replacement of targets (e.g. loss of genetic information, ataxia telangectasia being an hereditary ataxia in which an oxygen mediated chromosomal instability is apparent), (b) non-removal of unwanted materials (e.g. lipofuscin accumulation in brain and heart), (c) redox cycling, usually involving catalysis by trace-metal ions (e.g. some forms of Parkinsonism), (d) non-redox catalysis (e.g. toxicity in cardiac muscle or brain due to vanadium or aluminium respectively), (e) modification of ion transport (e.g. calcium ionophore or acrylamide induce histopathological changes in muscle, similar in some respects to those seen in Duchenne muscular dystrophy), (f) compromised defences (e.g. muscle and nerve become particularly susceptible to free radical damage after loss of the protective actions of vitamin E), and (g) amplification by inflammatory and immune responses (e.g. multiple sclerosis, reperfusion injury to brain and heart, and traumatic injury to nervous tissue). Unfortunately, a variety of therapeutic agents which might be expected to protect against almost every conceivable form of oxygen mediated damage have proved clinically ineffective in most of these disorders. The reasons for this will be explored with an emphasis on common features, differences, mechanisms, and potential therapeutic approaches.
Similar content being viewed by others
References
Kong S, Davison A: The relative effectiveness of ·OH, H2O2, O2, and reducing free radicals in causing damage to biomembranes. A study of radiation damage to erythrocyte ghosts using selective free radical scavengers. Biochem Biophys Acta 640:313–325, 1981
Davison AJ, Legault NA, Steele DW: Effect of 6-OHDA on polymerization of tubulin. Protection by superoxide dismutase, catalase, or anaerobic conditions. Biochem Pharmacol 35:1411–1417, 1986
Hatefi Y, Hanstein WG: Destabilization of membranes with chaotropic ions. Methods Enzymol 31:770–790, 1974
Panganamala RV, Cornwell DG: The effects of vitamin E on arachidonic acid metabolism. In B Lubin and LJ Machlin (eds). Vitamin E: Biochemical, Hematological, and Clinical Aspects. Ann NY Acad Sci 393:376–391, 1982
Davison AJ, Wilson BD, Belton P: Deterioration of axonal membranes induced by phenolic pro-oxidants. Roles of superoxide radicals and hydrogen peroxide. Biochem Pharmacol 33:3887–3891, 1984
Nohl H, Hegner D, Summer K-H: The mechanism of toxic action of hyperbaric oxygenation on the mitochondria of ratheart cells. Biochem Pharmacol 30:1753–1757, 1981
Miquel J, Economos AC, Fleming J, Johnson Jr, JE: Mitochondrial role in cell aging. Exp Gerontol 15:575–591, 1980
Halliwell B, Gutteridge JM, Blake D: Metal ions and oxygen radical reactions in human inflammatory joint disease. Philos Trans R Soc Lond [Biol] 311:659–671, 1985
Bennett T, Burnstock G, Cobb JLS, Malmfors T: An ultrastructural and histochemical study of the short-term effects of 6-OHDA on adrenergic nerves in the domestic fowl. Br J Pharmacol 38:802–809, 1970
Gee P: Mechanisms of cytotoxicity of 6-OHDA: roles of oxygen free radicals and the effects of scavengers. Ph.D. Thesis, Department of Kinesiology, Simon Fraser University, Burnaby, B.C., Canada, 1984
Gee P, Davison AJ: Effects of Scavengers of oxygen free radicals on the anaerobic oxidation of 6-OHDA by H2O2. Biochim Biophys Acta 838:183–190, 1985
Graham DG, Tiffany SM, Bell WR Jr., Gutknecht WF: Autoxidation versus covalent binding of quinones as the mechanism of toxicity of dopamine, 6-OHDA, and related compounds toward C1300 neuroblastoma cells in vitro. Mol Pharm 14:644–653, 1978
Bandy B, Davison AJ: Interactions between metals, ligands and oxygen in the autoxidation of 6-OHDA: mechanisms by which metal chelatinn enhances inhibition by superoxide dismutase. Arch Biochem Biophys 259:305–315, 1987
Biemond P, van Eijk HG, Swaak AJ, Koster JF: Iron mobilization from ferritin by superoxide derived from stimulated polymorphonuclear leukocytes. Possible mechanism in inflammation diseases. J Clin Invest 73:1576–1579, 1984
Stephenson M: Protein interaction with copper (II)/ascorbic acid/oxygen system: evidence for a non-site specific reaction. Rev Port Quim 27:250–253, 1985
Poirier J, Barbeau A: A catalyst function for MPTP in superoxide formation. Biochem Biophys Res Commun 131:1284–1289, 1985
Di Monte D, Sandy MS, Ekstrom G, Smith MT: Comparative studies on the mechanisms of paraquat and 1-methyl-4phenylpyridine (MPP+) cytotoxicity. Biochem Biophys Res Commun 137:303–309, 1986
Sershen H, Reith ME, Hashim A, Lajtha A: Protection against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine neurotoxicity by the antioxidant ascorbic acid. Neuropharmacology 24:1257–1259, 1985
Poirier J, Donaldson J, Barbeau A: The specific vulnerability of the substantia nigra to MPTP is related to the presence of transition metals. Biochem Biophys Res Commun 128:25–33, 1985
Smith MT, Ekstrom G, Sandy MS, Di Monte D: Studies on the mechanism of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine cytotoxicity in isolated hepatocytes. Life Sci 40:741–748, 1987
Martinovits G, Melamed E, Cohen O, Rosenthal J, Uzzan A: Systematic administration of antioxidants does not protect mice against the dopaminergic neurotoxicity of 1-methyl-4phenyl-1,2,5,6 tetrahydropyridine (MPTP). Neurosci Lett 69:192–197, 1986
Ramsay RR, Dadgar J, Trevor A, Singer TP: Energy-driven uptake of N-methyl-4-phenylpyridine by brain mitochondria mediates the neurotoxicity of MPTP. Life Sci 39:581–588, 1986
Nicklas WJ, Youngster SK, Kindt MV, Heikkila RE: MPTP, MPP+ and mitochondrial function. Life Sci 40:721–729, 1987
Anthony J, Fadl S, Mason C, Davison A, Berry J: Absorption, deposition and distribution of dietary aluminum in immature rats: effects of dietary vitamin D3 and food-borne chelating agent. J Environ Sci Health B21:191–205, 1986
Nayler RA, Sparrow MP: Inhibition of cycling and noncycling cross bridges in skinned smooth muscle by vanadate. Am J Physiol 250:C325–C332
Suh CK, Park SR, Ahn DS, Paik KS: Effects of vanadate on cellular Ca2+ movements in guinea pig papillary muscles. Yonsei Med J 28:23–30, 1987
Shimada T, Tsuji A, Shimamura K, Sunano S: Comparison of contractile effects of sodium vanadate and ouabain in vascular smooth muscles of guinea-pigs and rats. Nippon Heikatsukin Gakkai Zasshi 22:409–422, 1986
Wu Q: Redoxreactivity of vanadium toward cytochromecand oxygen. M.Sc. Thesis, Department of Kinesiology, Simon Fraser University, Burnaby, British Columbia, Canada, Dec. 1985
Tracey AJ, Gresser MJ: Interaction of vanadate with phenol and tyrosine: implications for effects of vanadate on systems regulated by tyrosine phosphorylation. Proc Nat Acad Sci USA 83:609–613, 1986
Domingo JL, Llobet JM, Corbella J: Protection of mice aginst the lethal effects of sodium metavanadate: a quantitative comparison of a number of chelating agents. Toxicol Lett 26:95–99, 1985
Steele DW, Davison AJ: Metal-stimulated autoxidation of 6-OHDA: involvement of dioxygen. Can Fed Biol See, 30th Annual Meeting, Winnipeg, Manitoba June 22–26, 1987
Steele DW, Davison AJ: Metal and metal-chelate catalyzed autoxidation of 6-OHDA: differential involvement of molecular oxygen. Pacific Slope Biochemical Conference, University of British Columbia, June 22–25, 1986
Mellgren RL: Calcium-dependent proteases: an enzyme system at cellular membranes. FASEB J 1:110–115, 1987
Kay J: Ca2+-activated proteinases, protein degradation and muscular dystrophy. Adv Exp Med Biol 167:519–531, 1984
Suzuki K: Calcium activated neutral protease: domain structure and activity regulation. Trends in Biochem Sci pp 103–105, March 1987
Yoshimura T, Tsujihata M, Satoh A, Mori M, Hazama R, Kinoshita N, Takashima H, Nagataki S: Ultrastructural study of calcium ionophore, A23187, on rat muscle. Acta Neuropathol 69:184–192, 1986
Jakobsen J, Sidenius P: Early and dose-dependent decrease of retrograde axonal transport in acrylamide-intoxicated rats. J Neurochem 40:447–454, 1983
Bisby MA, Redshaw JD: Acrylamide neuropathy: changes in the composition of proteins of fast axonal transport resemble those observed in regenerating axons. J Neurochem 48:924–928, 1987
Howland RD: Altered phosphorylation of rat neuronal cytoskeletal protein in acrylamide induced neuropathy. Brain Res 363:333–339, 1986
Rabbani N, Moses L, Anandavalli TE, Anandaraj MP: Calcium-activated neutral protease from muscle and platelets of Duchenne muscular dystrophy cases. Clin Chim Acta 143:163–168, 1984
Klamut HJ, Lin CH, Strickland KP: Calmodulin content and Ca-activated protease activity in dystrophic hamster muscles. Muscle Nerve 6:436–441, 1983
Leonard JP, Salpeter MM: Calcium-mediated myopathy at neuromuscular junctions of normal and dystrophic muscle. Exp Neurol 76:121–138, 1982
Tijoe SA, Hadjiconstantinuou M: MPTP induces lipofuscin accumulation in neuronal tissue. American Aging Association, 17th Annual National Meeting, Oct. 8–10, 1987
Murphy ME, Kehrer JP: Free radicals: a potential pathogenic mechanism in inherited muscular dystrophy. Life Sci 39:2271–2278, 1986
Parks DA, Granger DN: Ischemia-induced vascular changes: role of xanthine oxidase and hydroxyl scavengers. Am J Physiol 245:G285-G289, 1983
Doriguzzi C, Bertolotto A, Ganzit GP, Mongini T, Palmucci L: Ineffectiveness of allopurinol in Duchenne muscular dystrophy [letter]. Muscle Nerve 4:176–178, 1981
Griffiths RD, Cady EB, Edwards RH, Wilkie DR: Muscle energy metabolism in Duchenne dystrophy studied by 31P-NMR: controlled trials show no effect of allopurinol or ribose. Muscle Nerve 8:760–767, 1985
Edwards RH, Jones DA, Jackson MJ: An approach to treatment trials in muscular dystrophy with particular reference to agents influencing free radical damage. Med Biol 62:143–147, 1984
Pappenheimer AM, Goettsch MA: A cerebellar disorder in chicks, apparently of nutritional origin. J Exp Med 53:11–16, 1931
Milhorat AT. Therapy in muscular dystrophy. Med Ann DC 23:15–22, 1954
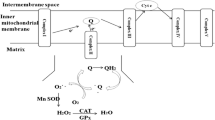
Haugaard N: Cellular mechanisms of oxygen toxicity. Physiol Rev 48:311–373, 1968
Haeger K: Long-term study of α tocopherol in intermittent claudication. In B Lubin and LJ Machlin (eds). Vitamin E: Biochemical, Hematological, and Clinical Aspects. Ann NY Acad Sci 393:369–375, 1982
Barness LA: Progressive neuromuscular disease in children with chronic cholestasis and vitamin E deficiency: clinical and muscle biopsy findings and treatment with α-tocopherol. In B Lubin and LJ Machlin (eds). Vitamin E: Biochemical, Hematological, and Clinical Aspects. Ann NY Acad Sci 393:84–95, 1982
Muller DPR, Lloyd JK: Effect of large oral doses of vitamin E on the neurological sequelae of patients with abetalipoproteinemia. In B Lubin and LJ Machlin (eds). Vitamin E: Biochemical, Hematological, and Clinical Aspects. Ann NY Acad Sci 393:133–144, 1982
Kretzer FL, Hittner HM, Johnson AT, Mehra RS, Godio LB: Vitamin E and retrolental fibroplasia: ultrastructural support of clinical efficacy. In B Lubin and LJ Machlin (eds). Vitamin E: Biochemical, Hematological, and Clinical Aspects. Ann NY Acad Sci 393:145–167, 1982
Johnson L, Schaffer D, Quinn G, Goldstein D, Mathis MJ, Otis C, Boggs TR Jr: Vitamin E supplementation and the retinopathy of prematurity. In B Lubin and LJ Machlin (eds). Vitamin E: Biochemical, Hematological, and Clinical Aspects. Ann NY Acad Sci 393:473–495, 1982
Davison AJ, Kettle AJ, Fatur DJ: Mechanism of inhibition of catalase by ascorbate. Roles of active oxygen species, copper and semidehydroascorbate. J Biol Chem 261:1193–1200, 1986
Bray RC, Cockle SA, Fielden EM, Roberts PB, Rotilio G, Calabrese L: Reduction and inactivation of superoxide dismutase by hydrogen peroxide. Biochem J 139:43–58, 1974
Yoshida S, Busto R, Santiso M, Ginsberg MD: Brain lipid peroxidation induced by postischemic reoxygenation in vitro: effect of vitamin E. J Cereb Blood Flow Metab 4:466–469, 1984
Anderson DK, Demediuk P, Saunders RD, Dugan LL, Means ED, Horrocks LA: Spinal cord injury and protection. Ann Emerg Med 14:816–821, 1985
Anderson DK, Means ED: Iron-induced lipid peroxidation in spinal cord: protection with mannitol and methylprednisolone. J Free Radic Biol Med 1:59–64, 1985
Anderson DK, Means ED, Waters TR, Green ES: Microvascular perfusion and metabolism in injured spinal cord after methylprednisolone treatment. J Neurosurg 56:106–113, 1983
Braughler JM, Hall ED, Means ED, Waters TR, Anderson DK: Evaluation of an intensive methylprednisolone sodium succinate dosing regimen in experimental spinal cord injury. J Neurosurg 67:102–105, 1987
Demediuk P, Saunders RD, Clendenon NR, Means ED, Anderson DK, Horrocks LA: Changes in lipid metabolism in traumatize spinal cord. Prog Brain Res 63:211–226, 1985
Demediuk P, Saunders RD, Anderson DK, Means ED, Horrocks LA: Membrane lipid changes in laminectomized and traumatized cat spinal cord. Proc Natl Acad Sci USA 82:7071–7075, 1985
Hall ED, Braughler JM: Role of lipid peroxidation in posttraumatic spinal cord degeneration: a review. Cent Nerv Syst Trauma 3:281–294, 1986
Kontos HA, Povlishock JT Oxygen radicals in brain injury. Cent Nerv Syst Trauma 3:257–263, 1986
Au AM, Chan PH, Fishman RA: Stimulation of phospholipase A2 activity by oxygen-derived free radicals in isolated brain capillaries. J Cell Biochem 27:449–453, 1985
Cao W, Carney JM, Duchon A, Floyd RA, Chevion M: Direct evidence for the role of oxygen free radicals in ischemia-reperfusion induced brain damage. American Aging Association, 17th Annual National Meeting, Oct. 8–10, 1987
Woodruff T, Blake DR, Freeman J, Andrews FJ, Salt P, Lunec J: Is chronic synovitis an example of reperfusion injury? Ann Rheum Dis 45:608–611, 1986
Graham DG: Catecholamine toxicity: A proposal for the molecular pathogenesis of manganese neurotoxicity and Parkinson's disease. Neurotoxicity 5:83–96, 1984
Howland RD: Biochemical studies of acrylamide neuropathy. Neurotoxicology 6:7–15, 1985
Porubek DJ, Rundgren M, Harvison PJ, Nelson SD, Moldeus P: Investigation of mechanisms of acetaminophen toxicity in isolated rat hepatocytes with the acetaminophen analogues 3,5-dimethylacetaminophen and 2,6-dimethylacetaminophen. Mol Pharmacol 31:647–653, 1987
Jackson MJ, Jones DA, Edwards RH: Techniques for studying free radical damage in muscular dystrophy. Med Biol 62:135–138, 1984
Mizuno Y Superoxide dismutase activity in early stages of development in normal and dystrophic chickens. Life Sci 34:909–914, 1984
Matkovics B, Laszlo A, Szabo L: A comparative study of superoxide dismutase, catalase and lipid peroxidation in red blood cells from muscular dystrophy patients and normal controls. Clin Chim Acta 118:289–292, 1982
Perkins RC, Beth AH, Wilkerson LS, Serafin W Dalton LR, Park CR, Park JH: Enhancement of free radical reduction by elevated concentrations of ascorbic acid in avian dystrophic muscle. Proc Natl Acad Sci USA 77:790–794, 1980
Mechler F, Imre S, Dioszeghy P: Lipid peroxidation and superoxide dismutase activity in muscle and erythrocytes in Duchenne muscular dystrophy. J Neurol Sci 63:279–283, 1984
Burri BJ, Chan SG, Berry AJ, Yarnell SK: Blood levels of superoxide dismutase and glutathione peroxidase in Duchenne muscular dystrophy. Clin Chim Acta 105:249–255, 1980
Becker Y, Shaham M, Tabor E, Shiloh Y Ataxia-telangiectasia — a human autosomal recessive disorder predisposing to cancer. In G Giraldo and E Beth (eds). The Role of Viruses in Human Cancer, Volume II. New York, Elsevier Science Publishers, pp 397–496, 1984
Cornforth MN, Bedford JS: On the nature of a defect in cells from individuals with ataxia-telangiectasia. Science 227:1589–1591, 1985
Joenje H, Nieuwint AW Taylor AM, Harnden DG: Oxygen toxicity and chromosomal breakage in ataxia telangiectasia. Carcinogenesis 8:341–344, 1987
Sheridan RB 3d, Huang PC: Superoxide dismutase and catalase activities in ataxia telangiectasia and normal fibroblast cell extracts. Mutat Res 61:381–386, 1979
Shiloh Y, Tabor E, Becker Y Abnormal response of ataxiatelangiectasia cells to agents that break the deoxyribose moiety of DNA via a targeted free radical mechanism. Carcinogenesis 4:1317–1322, 1983
Munoz-Garcia D, Pendlebury WW Kessler JB, Perl DP: An immunocytochemical comparison of cytoskeletal proteins in aluminum-induced and Alzheimer-type neurofibrillary tangles. Acta Neuropathol 70:234–238, 1986
Packer L: Vitamin E, physical exercise and tissue damage in animals. Medical Biology 62:105–109, 1984
Gohil K, Packer L, de Lumen B, Brooks GA, Terblanche SE: Vitamin E deficiency and vitamin C supplements: exercise and mitochondrial oxidation. J Appl Physiol 60:1986–1991, 1986
Goto I, Shinno N, Kuroiwa Y: Proteolytic enzyme activities, in mononuclear cells and granulocytes of patients with various neurological disorders. J Neurol Sci 59:323–329, 1983
Gomez CM, Wollmann RL, Richman DP: Induction of the morphologic changes of both acute and chronic experimental myasthenia by monoclonal antibody directed against acetylcholine receptor. Acta Neuropathol 63:131–143, 1984
Koziner B, Bloch KJ, Perlo VP: Distribution of peripheral blood latex-ingesting cells, T cells, and B cells in patients with myasthenia gravis. Ann NY Acad Sci 274:411–420, 1976
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Davison, A., Tibbits, G., Shi, Z. et al. Active oxygen in neuromuscular disorders. Mol Cell Biochem 84, 199–216 (1988). https://doi.org/10.1007/BF00421055
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00421055