Abstract
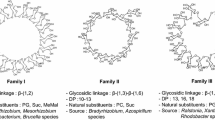
Gel-filtration and thin layer chromatography of low molecular weight carbohydrates from culture filtrates of Agrobacterium radiobacter, Isolate II, have shown, that next to the neutral β-1,2-glucan fraction a major acidic fraction was present which was found to be glycerophosphorylated cyclic β-1,2-glucans. Re-examination of cyclic β-1,2-glucan preparations which had been obtained by extraction of Rhizobium cells with hot phenol-water also showed these acidic modified β-1,2-glucans to be present. Cyclic β-1,2-glucans from R. leguminosarum (9 strains) and of R. phaseoli (1 strain) had ring size distribution with degrees of polymerisation (DPs) of 19 and 20 as major ring sizes of which a minor part was glycerophosphorylated; β-1,2-glucans of R. trifolii (3 strains) had ring sizes with DPs measuring 19–22 as prominent components which were largely unsubstituted, and R. meliloti (7 strains) had β-1,2-glucans with ring size distributions extending to still higher DPs of 19–25 of which the major part appeared to be glycerophosphorylated.
Similar content being viewed by others
References
Abe M, Amemura A & Higashi S (1982) Studies on cyclic β-1,2-glucans obtained from periplasmic space of Rhizobium trifolii cells. Plant & Soil 64: 315–324
Amemura A, Hisamatsu M, Mitani H & Harada T (1983) Cyclic (1»2)-β-D-glucan and the octasaccharide repeating units of extracellular acidic polysaccharides produced by Rhizobium. Carbohydr. Res 114: 277–285
Amemura A, Footrakul P, Koizumi K, Utamura T & Taguchi H (1985) Isolation of (1→2)-β-D-glucans from tropical strains of Rhizobium and determination of their degrees of polymerisation. J. Ferm. Technol. 63: 115–121
Amemura A & Cabrero-Crespo J (1986) Extracellular oligosaccharides and low Mrpolysaccharides containing (1→2)-β-D-glucosidic linkages from strains of Xanthomonas, Escherichia coli and Klebsiella pneumoniae. J. Gen. Microbiol. 132: 2443–2452
Batley M, Redmond JW, Djordjevic SP & Wolfe BG (1987) Characterization of glycerophosphorylated cyclic β-1,2-glucans from a fast-growing Rhizobium species. Bioch. Biophys. Acta 901: 119–126
Benincasa M, Cartoni GP, Coccioli F, Rizzo R & Zevenhuizen LPTM (1987) High-performance liquid chromatography of cyclic β-(1→2)-D-glucans (cyclosophoraoses) produced by Rhizobium meliloti and Rhizobium trifolii. J. Chromatography 393: 263–271
Kennedy EP & Rumley MK (1988) Osmotic regulation of biosynthesis of membrane-derived oligosaccharides in Escherichia coli. J. Bact. 170: 2457–2461
Madsen NB (1962) Periodation of the glucans of Agrobacterium tumefaciens with recovery of cleavage products. Anal. Biochem. 4: 509–511
McIntire FC, Peterson WH & Riker AJ (1942) A polysaccharide produced by the crown-gall organism. J. Biol. Chem. 143: 491–496
Miller KJ, Kennedy EP & Reinhold VN (1986) Osmotic adaptation by Gram-negative bacteria: possible role for periplasmic oligosaccharides. Science 231: 48–51
Putman EW, Potter AL, Hodgson R & Hassid WZ (1950) The structure of the crown-gall polysaccharide. J. Am. Chem. Soc. 72: 5024–5026
Schneider JE, Reinhold V, Rumley MR & Kennedy EP (1979) Structural studies of the membrane-derived oligosaccharides of Escherichia coli. J. Biol. Chem. 254: 10135–10138
Schulman H & Kennedy EP (1979) Localisation of membranederived oligosaccharides in the outer envelope of Escherichia coli and their occurrence in other Gram-negative bacteria. J. Bact. 137: 686–688
Trevelyan WC & Harrison JS (1952) Studies on yeast metabolism. I. Fractionation and microdetermination of cell carbohydrates. Biochem. J. 50: 298–310
York WS, McNeil M, Darvil AG & Albersheim P. (1980) Hostsymbiont interactions VIII. β-2-Linked glucans secreted by fast-growing species of Rhizobium. J. Bact. 142: 243–248
Zevenhuizen LPTM & Scholten-Koerselman HJ (1979) Surface carbohydrates of Rhizobium. I. β-1,2-Glucans. Antonie van Leeuwenhoek 45: 165–175
Zevenhuizen LPTM, Scholten-Koerselman I & Posthumus MA (1980) Lipopolysaccharides of Rhizobium. Arch. Microbiol. 125: 1–8
Zevenhuizen LPTM (1981) Cellular glycogen, β-1,2-glucan, poly-β-hydroxybutyric acid and extracellular polysaccharides in fast-growing species of Rhizobium. Antonie van Leeuwenhoek 47: 481–497
Zevenhuizen LPTM & VanNeervenARW (1983) (1→2)-β-D-Glucan and acidic oligosaccharides produced by Rhizobium meliloti. Carbohydr. Res. 118: 127–134
Zevenhuizen LPTM (1986) Selective synthesis of polysaccharides by Rhizobium trifolii, strain TA-1. FEMS Microbiol. Letters 35: 43–47
Zevenhuizen LPTM (1989) Diversity of polysaccharide synthesis within a restricted group of bacteria. In: CrescenziV, DeaICM, PaolettiS, StivalaSS & SutherlandIW (Eds) Industrial Polysaccharides. Gordon & Breach, Science Publishers, New York, (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zevenhuizen, L.P.T.M., van Veldhuizen, A. & Fokkens, R.H. Re-examination of cellular cyclic β-1,2-glucans of Rhizobiaceae: Distribution of ring sizes and degrees of glycerol-1-phosphate substitution. Antonie van Leeuwenhoek 57, 173–178 (1990). https://doi.org/10.1007/BF00403952
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00403952