Summary
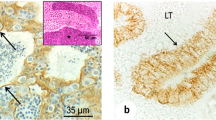
In this paper we present evidence for the presence of actin-related junctions between neighboring Sertoli cells and between Sertoli cells and spermatids in the testis of the guppy (Poecilia reticulata). In the guppy, spermatogenesis occurs in spermatocysts that are lined by a simple squamous to cuboidal epithelium formed of Sertoli cells. At a certain stage of differentiation, elongate spermatids occur in Sertoli cell recesses in the apical surface of Sertoli cells. When evaluated by electron microscopy, junctions occur between Sertoli cells and spermatids situated in the recesses. In these regions, obvious linkages occur between the plasma membrane of Sertoli cell recesses and the adjacent spermatids. Moreover, large concentrations of microfilaments occur in the Sertoli cell cytoplasm immediately underlying the crypts. Also, junctional complexes are apparent between neighboring Sertoli cells near the apical surface of the epithelium. These complexes consist of microfilament-related components (probably contributing to both tight and adhesion junctions), which occur closest to the lumen, and intermediate-filament related desmosomes, which occur more basally. In fixed frozen sections of guppy testis, probes for filamentous actin (rhodamine phalloidin) and myosin II (polyclonal antisera raised against human platelet myosin II) react with function regions between neighboring Sertoli cells and between Sertoli cells and spermatids. We conclude that actin-related junctions occur at both these sites and that the actin networks have contractile properties because they contain myosin II.
Similar content being viewed by others
References
Bergmann M, Schindelmeiser J, Greven H (1984) The blood-testis barrier in vertebrates having different testicular organization. Cell Tissue Res 238:145–150
Billard R (1970) La spermatogenese de Poecilia reticulata. III. —Ultrastructure des cellules de Sertoli. Ann Biol Anim Biochim Biophys 10:37–50
Billard R (1984) Ultrastructural changes in the spermatogonia and spermatocytes of Poecilia reticulata during spermatogenesis. Cell Tissue Res 237:219–226
Billard R (1986) Spermatogenesis and spermatology of some teleost fish species. Reprod Nutr Develop 26:877–920
Boller K, Vestweber D, Kemler R (1985) Cell-adhesion molecule uvomorulin is localized in the intermediate junctions of adult intestinal epithelial cells. J Cell Biol 100:327–332
Byers S, Pelletier R-M, Suarez-Quian C (1993) Sertoli cell junctions and the seminiferous epithelium barrier. In: Russell LD, Griswald MD (eds) The Sertoli cell. Cache River Press, Clearwater, Florida, pp 431–446
Drenckhahn D, Dermietzel R (1988) Organization of the actin filament cytoskeleton in the intestinal brush border: a quantitative and qualitative immunoelectron microscope study. J Cell Biol 107:1037–1048
Farquhar MG, Palade GE (1963) Junctional complexes in various epithelia. J Cell Biol 17:375–409
Grier HJ (1975) Aspects of germinal cyst and sperm development in Poecilia latipinna (Teleostei: Poeciliidae). J Morphol 146:229–250
Grier HJ (1993) Comparative organization of Sertoli cells including the Sertoli cell barrier. In: Russell LD, Griswald MD (eds) The Sertoli cell. Cache River Press, Clearwater, Florida, pp 703–739
Gumbiner B (1992) Cell adhesion molecules in epithelia. In: Cereijido M (ed) Tight junctions. CRC Press, Boca Raton, Florida, pp 91–104
Hirano S, Nose A, Hatta K, Kawakami A, Takeichi M (1987) Calcium-dependent cell-cell adhesion molecules (cadherins): subclass specificities and possible involvement of actin bundles. J Cell Biol 105:2501–2510
Hirokawa N, Keller III TCS, Chasan R, Mooseker MS (1983) Mechanism of brush border contractility studies by the quick-freeze, deep-etch method. J Cell Biol 96:1325–1336
Madara JL (1987) Intestinal absorptive cell tight junctions are linked to the cytoskeleton. Am J Physiol 253:C171–175
Madara JL (1992) Relationships between the tight junction and the cytoskeleton. In: Cereijido M (ed) Tight junctions. CRC Press, Boca Raton, Florida, pp 105–119
Marcaillou C, Szollosi A (1980) The “blood-testis” barrier in a nematode and a fish: a generalizable concept. J Ultrastruct Res 70:128–136
Mooseker MS (1985) Organization, chemistry, and assembly of the cytoskeletal apparatus of the inestinal brush border. Ann Rev Cell Biol 1:209–241
Pfeiffer DC, Vogl AW (1993) Ectoplasmic (“junctional”) specializations in Sertoli cells of the rooster and turtle: Evolutionary implications. Anat Rec 235:33–50
Philp NJ, Nachmias VT (1985) Components of the cytoskeleton in the retinal pigmented epithelium of the chick. J Cell Biol 101:358–362
Pudney J (1993) Comparative cytology of the non-mammalian vertebrate Sertoli cell. In: Russell LD, Griswald MD (eds) The Sertoli cell. Cache River Press, Clearwater, Florida, pp 611–657
Russell LD (1977) Observations on rat Sertoli ectoplasmic (‘junctional’) specializations in their association with germ cells of the rat testis. Tissue Cell 9:475–498
Sprando RL, Russell LD (1987) A comparative study of Sertoli cell ectoplasmic specializations in selected non-mammalian vertebrates. Tissue Cell 19:479–493
Vogl AW, Pfeiffer DC, Redenbach DM, Grove BD (1993) The Sertoli cell cytoskeleton. In: Russell LD, Griswald MD (eds) The Sertoli cell. Cache River Press, Clearwater, Florida, pp 39–86
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pfeiffer, D.C., Vogl, A.W. Actin-related intercellular junctions in the germinal compartment of the testis in Poecilia reticulata (Teleostei, Poeciliidae). Zoomorphology 114, 177–184 (1994). https://doi.org/10.1007/BF00403265
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00403265