Summary
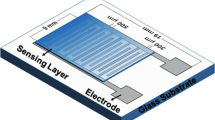
A galvanic sensor for monitoring nitrogen dioxide was developed by using a poly(ethylene oxide) complex of silver trifluoromethanesulphonate electrolyte. The sensor, which is expressed as Au/P(EO)4.5 AgCF3SO3/Ag, is a small disk (i.d. 13 mm). The polymeric electrolyte film was made by casting the mixture of acetonitrile solutions of both P(EO) (MW 6×105) and AgCF3SO3. The working electrode was made by sputtering of gold in argon. The thicknesses of the desposited gold, polymeric electrolyte film and silver are 25 nm, 30 μm and 1 mm, respectively. When the sample gas containing nitrogen dioxide impinges at 20 ml min−1 on the gold cathode, the current flowing in the external circuit was linearly related to the concentration of nitrogen dioxide from 20 ppb to about 10 ppm. The current efficiency of the cell was 0.051%. The cell's response time was about 2 min for 0.5 ppm of nitrogen dioxide.
Similar content being viewed by others
References
Janata J, Huber RJ (1985) Solid state chemical sensors. Academic Press
Schuetzle D, Hammerle R (1986) Fundamentals and applications of chemical sensors. Am Chem Soc
Proceedings of the Fourth International Conference on Solidstate Sensors and Actuators (1987) Institute of Electrical Engineers of Japan
Janata J (1989) Principles of chemical sensors. Plenum
Suzuki S, Nagashima K (1982) Anal Chim Acta 144:261
Nagashima K, Meguro K, Suzuki S, Hobo T (1988) Bunseki Kagaku 37:400
Fenton BE, Parker JM, Wright PV (1973) Polymer 14:589
Armand M, Chabagno J, Duclot M (1979) Fast ion transport in solids. North-Holland, New York, p 131
Rietman EA, Kaplan ML, Cava RJ (1987) Solid State Ion 25:41
Blonsky PM, Shriver DF (1984) J Am Chem Soc 106:6854
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nagashima, K., Meguro, K. & Hobo, T. A galvanic gas sensor using poly (ethylene oxide) complex of silver trifluoromethane sulphonate electrolyte. Fresenius J Anal Chem 336, 571–574 (1990). https://doi.org/10.1007/BF00331419
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00331419