Abstract
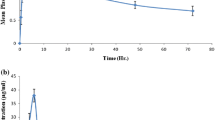
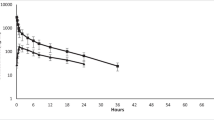
The plasma kinetics of parathion were studied in rabbits after i.v. administration of a dose of 1.5 mg/kg and oral administration of 3 mg/kg. The time course of parathion plasma levels administered intravenously followed a three-compartment kinetic model statistically, whereas when administration was oral, the optimum kinetic model proved to be two-compartmental. The process of the absorption of parathion is very fast with a mean value for the absorption constant (ka) of 33±15.41 h−1. The slow disposition half-lives for i.v. and oral administration had mean values of 5.08±3.08 and 1.08±0.27h, respectively. From the values established for the parameters defining the distribution process the wide accessibility of parathion to the different body organs and tissues may be seen. Although the compound has a high elimination constant, this process is not limiting to distribution.
Similar content being viewed by others
References
Ahdaya SM, Monroe RJ, Guthrie FE (1981) Absorption and distribution of intubated insecticides in fasted mice. Pestic Biochem Physiol 16: 38–46
Antunes-Madeira MC, Madeira VMC (1984) Partition of parathion in synthetic and native membranes. Biochim Biophys Acta 778: 49–56
Appleton HT, Nakatsugawa T (1977) The toxicological significance of paraoxon deethylation. Pestic. Biochem Physiol 7: 451–465
Baker SR, McGilliard AD, Dahm PA (1979) Uptake, clearance and metabolism of parathion by perfused whole pig livers. Pestic Biochem Physiol 11: 20–32
Braeckman RA, Audenaert F, Willems JL, Belpaire FM, Bogaert MG (1983) Toxicokinetics of methyl parathion and parathion in the dog after intravenous and oral administration. Arch Toxicol 54: 71–82
Brown RD, Manno JE (1978) ESTRIP, a basic computer program for obtaining initial polyexponential parameter estimates. J Pharm Sci 57: 1687–1691
Chadwick RW, Copeland MF, Froehlich R, Cooke N, Whitehouse DA (1984) Interaction between γ-hexachlorocyclohexane and the gastrointestinal microflora and their effect on the absorption, biotransformation and excretion of parathion by the rat. J Agric Food Chem 32: 755–759
De Broe ME, Bismuth C, De Groot G, Heath A, Okonek S, Ritz DR, Verpooten GA, Volans GN, Widdop B (1986) Haemoperfusion: a useful therapy for a severely poisoned patient? Hum Toxicol 5: 11–14
De Potter M, Muller R, Willems J (1978) A method for the determination of some organophosphorus insecticides in human serum. Chromatographia 11: 220–222
Fournier E, Sonnier M, Dally S (1978) Detection and assay of organophosphate pesticides in human blood by gas chromatography. Clin Toxicol 12: 457–462
Gibaldi M, Perrier D (1982) Farmacocinética (Plá-Delfina JM y Moreno Dalman, trads.) Ed. Reverté, Barcelona, pp 334
Knaak JB, Yee K, Ackerman CR, Zweig G, Fry DM, Wilson BW (1984) Percutaneous absorption and dermal dose -cholinesterase response studies with parathion and carbaryl in the rat. Toxicol Appl Pharmacol 76: 252–263
Okonek S (1978) Elimination of parathion by hemoperfusion in severe E 605 forte® intoxication in vivo. Naunyn-Schmiedeberg's Arch Pharmacol 302: R 66
Okonek S, Kilbinger H (1974) Determination of acetylcholine, nitrostigmine and acetyl cholinesterase activity in four patients with severe nitrostigmine (E 605 forte®) intoxication. Arch Toxicol 32: 97–108
Pentz R, Brunn J (1979) Hämoperfusion mit absorbenharz XAD-4 bei einer extrem schweren parathion-vergiftung. Arch Toxicol 42: 311–315
Roan CC, Morgan DP, Cook N, Paschal EH (1969) Blood Cholinesterases, serum parathion concentrations and urine p-nitrophenol concentrations in exposed individuals. Bull Environ Contam Toxicol 4: 362–369
Sheiner BL (1983) ELSFIT: program for extend least squares fit to individual pharmacokinetic data. Division of clinical pharmacology. University of California of San Francisco, USA
Sultatos LG, Basker KM, Shao M, Murphy SD (1984) The interaction of the phosphorothionate insecticides chlorpyrifos and parathion and their oxygen analogues with bovine serum albumin. Mol Pharmacol 26: 99–104
Vukovic RA, Triolo AJ, Coon JM (1969) Rapid method for detection of parathion in plasma by electron capture gas chromatography without prior cleanup. J Agric Food Chem 17: 1190–1191
Weitman SD, Vodicnik MJ, Lech JJ (1983) Influence of pregnancy on parathion toxicity and disposition. Toxicol Appl Pharmacol 71: 215–224
Yamaoka P, Nakagawa T, Uno T (1978) Application of Akaike's information criterion in the evaluation of linear pharmacokinetics equation. J Pharmacokin Biopharm 6: 165–175
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peña-Egido, M.J., Rivas-Gonzalo, J.C. & Mariño-Hernandez, E.L. Toxicokinetics of parathion in the rabbit. Arch Toxicol 61, 196–200 (1988). https://doi.org/10.1007/BF00316634
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00316634