Summary
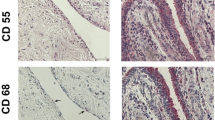
Primary cultures of adherent rheumatoid synovial cells (ASC) are comprised of variable proportions of fibroblasts, macrophages and stellate cells (activated fibroblasts). These cultures were shown to produce the metalloproteinases stromelysin-1 (MMP-3), gelatinase A (MMP-2) and gelatinase B (MMP-9) by Western blotting and zymography techniques. Immunolocalisation studies showed that MMP-3 was mainly produced by the fibroblastic cells whereas MMP-9 was restricted to macrophages (CD68 positive). Subcultured synovial fibroblasts, devoid of macrophages, did not produce MMP-9 as judged by zymography and immunolocalisation; but when stimulated with phorbol myristate acetate and interleukin-1α both MMP-9 and MMP-3 were co-expressed. These ‘activated’ fibroblasts assumed a dendritic or stellate morphology, which in localisation studies was usually associated with enhanced enzyme production. Immunolocalisation studies of rheumatoid synovial tissue showed that relatively few cells were positive for MMP-3 and MMP-9. Localisation of MMP-9 corresponded to a proportion of macrophages positive for the CD68 marker throughout the synovial tissue. MMP-3 localisation was not associated with the macrophage marker, but was observed in both the synovial lining layer and deeper stromal locations. Widespread distribution of both enzymes was not observed in fresh tissues, but this increased in tissues subjected to short-term explant cultures. Thus, both in vitro and in vivo studies indicated that synovial fibroblasts or B-cells are effective producers of MMP-3 whereas macrophages elaborate MMP-9, observations that demonstrated different metalloproteinase phenotypes under similar environmental conditions.
Similar content being viewed by others
References
Matrisian LM (1990) Metalloproteinases and their inhibitors in matrix remodelling. Trends Genet 6:121–125
Woessner JF (1991) Matrix metalloproteinases and their inhibitors in connective tissue remodelling. FASEB J 5:2145–2154
Nagase H, Barrett AJ, Woessner JM (1992) Nomenclature and glossary of the matrix metalloproteinases. Matrix [Suppl] 1:421–424
Woolley DE, Crossley MJ, Evanson JM (1977) Collagenase at sites of cartilage erosion in the rheumatoid joint. Arthritis Rheum 20:1231–1239
Okada Y, Nagase H, Harris ED Jr (1986) A metalloproteinase from human rheumatoid synovial fibroblasts that digests connective tissue matrix components. Purification and characterization. J Biol Chem 261:14245–14255
Okada Y, Shirimer M, Tanaka O, Naka K, Kimura A, Nakarishi I, Bayliss MT, Iwata K, Nagase H (1992) Localisation of matrix metalloprotenase 3 (stromelysin) in osteoarthritic cartilage and synovium. Lab Invest 66:680–686
McCachren SS (1991) Expression of metalloproteinases and metalloproteinase inhibitor in human arthritic synovium. Arthritis Rheum 34:1085–1093
Burmester GR, Dimitriu-Bona A, Waters SJ, Winchester RJ (1983) Identification of three major synovial lining cell populations by monoclonal antibodies directed to Ia antigens and antiens associated with monocytes/macrophages and fibroblasts. Scand J Immunol 17:69–82
Gadher SJ, Woolley DE (1987) Comparative studies of adherent rheumatoid synovial cells in primary culture: characterisation of the dendritic (stellate) cell. Rheumatol Int 7:13–22
Woolley DE, Brinckerhoff CE, Mainardi CL, Vater CA, Evanson JM, Harris ED Jr (1979) Collagenase production by rheumatoid synovial cells: morphological and immunohistochemical studies of the dendritic cells. Ann Rheum Dis 38:262–270
Okada Y, Takeuchi N, Tomita K, Nakanishi I, Nagase H (1989) Immunolocalization of matrix metalloproteinase 3 (stromelysin) in rheumatoid synovioblasts (B cells): correlation with rheumatoid arthritis. Ann Rheum Dis 48:645–653
Okada Y, Gonoji Y, Nahanishi I, Nagase H, Hayakawa T (1990) Immunohistochemical demonstration of collagenase and tissue inhibitor of metalloproteinases (TIMP) in synovial lining cells of rheumatoid synovium. Virchows Arch [b] 59:305–312
Unemori EN, Hibbs MS, Amento EP (1991) Constitutive expression of a 92 kDa gelatinase (type V collagenase) by rheumatoid synovial fibroblasts and its induction in normal human fibroblasts by inflammatory cytokines. J Clin Invest 88:1656–1662
Hibbs MS (1992) Expression of 92 kDa phagocyte elastinase by inflammatory and connective tissue cells. Matrix [Suppl] 1:51–57
Murphy G, Hembry RM, McGarrity AM, Reynolds JJ, Henderson B (1989) Gelatinase (type IV collagenase) immunolocalization in cells and tissues: use of an antiserum to rabbit bone gelatinase that identifies high and low Mr forms. J Cell Sci 92:487–496
Senior RM, Griffin GL, Fliszar CJ, Shapiro SD, Goldberg GI, Welgus HG (1991) Human 92-and 72-kilodalton type IV collagenases are elastases. J Biol Chem 266:7870–7875
Murphy G, Cooket MI, Ward RV, Docherty AJP (1991) Matrix metalloproteinase degradation of elastin, type IV collagen and proteoglycan. A comparison of the activities of 92 kDa and 72 kDa gelatinases, stromelysins-1 and-2 and punctuated metalloproteinases (PUMP). Biochem J 277:277–279
Arnett FC, Edworthy SM, Bloch DA (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:349–354
Dayer JM, Krane SM, Russell RGG, Robinson DR (1976) Production of collagenase and prostaglandins by isolated adherent rheumatoid synovial cells. Proc Natl Acad Sci USA 73:945–949
Morodomi T, Ogata Y, Sasaguri Y, Morimatsu M, Nagase H (1992) Purification and characterisation of matrix metalloproteinase 9 from U937 monocytic leukaemia and HT1080 fibrosarcoma cells. Biochem J 285:603–611
Ogata Y, Pratta MA, Nagase H, Arner EC (1992) Matrix metalloproteinase 9 (92 kDa gelatinase/type IV collagenase) is induced in rabbit articular chondrocytes by cotreatment with interleukin 1β and α protein kinase C activator. Exp Cell Res 201:245–249
Okada Y, Morodomi T, Enghild JJ, Suzuki K, Yasui A, Nakanishi I, Salvesen G, Nagase H (1990) Matrix metalloproteinase 2 from human rheumatoid synovial fibroblasts. Purification and activation of the precursor and enzymic properties. Eur J Biochem 194:721–730
Hipps DS, Hembry RM, Docherty AJP, Reynolds JJ, Murphy G (1991) Purification and characterisation of human 72 kDa gelatinase (type IV collagenase). Biol Chem Hoppe Seyler 372:287–296
Lefebre V, Peeters-Joris C, Vaer G (1991) Production of gelatin-degrading matrix metalloproteinases (type IV collagenases) and inhibitors by articular chondrocytes during their dedifferentiation by serial subcultures and under stimulation by interleukin-1 and tumor necrosis factor-α. Biochim Biophys Acta 1094:8–18
Ogata Y, Enghild JJ, Salvesen G, Nagase H (1992) Activation of the precursor for the human matrix metalloproteinase-9 by matrix metalloproteinase 3 (Stromelysin). J Biol Chem 267:3581–3584
Dayer JM, Beutler B, Cerami A (1985) Cachectin/tumor necrosis factor stimulates collagenase and prostaglandin E2 production by human synovial cells and dermal fibroblasts. J Exp Med 162:2163–2168
Dayer JM, de Rochemonteix B, Burrus B, Demczuk S, Dinarello CA (1986) Human recombinant interleukin-1 stimulates collagenase and prostaglandin E2 production by human synovial cells. J Clin Invest 77:645–648
MacNaul KL, Chartrain N, Lark M, Tocci MJ, Hutchinson NI (1990) Discoordinate expression of stromelysin, collagenase and tissue inhibitor of metalloproteinases-1 in rheumatoid human synovial fibroblasts. Synergistic effects of interleukin-1 and tumor necrosis factor-α on stromelysin expression. J Biol Chem 265:17238–17245
Feldmann M, Brennan FM, Chantry D, Haworth C, Turner M, Abney E, Buchan G, Barrett K, Barkley D, Chu A, Field M, Maini RN (1990) Cytokine production in the rheumatoid joint. Ann Rheum Dis 49:480–486
Brennan FM, Field M, Chu CQ, Feldmann M, Maini RN (1991) Cytokine expression in rheumatoid arthritis. Br J Rheumatol 30:[Suppl] 1:76–80
Nakajima H, Hiyama Y, Tsukada W, Warabi H, Uchida S, Hirose S (1990) Effects of interferon gamma on cultured synovial cells from patients with rheumatoid arthritis. Inhibition of cell growth, prostaglandin E2 and collagenase release. Ann Rheum Dis 49:512–516
Overall CM, Wrana JL, Sodek J (1989) Independent regulation of collagenase, 72 kDa progelatinase and metalloendoproteinase inhibitor expression in human fibroblasts by transforming growth factor beta. J Biol Chem 264:1860–1869
Alvaro-Garcia JM, Zvaifler NJ, Firestein GS (1990) Cytokines in chronic inflammatory arthritis. V. Mutual antagonism between interferon-gamma and tumor necrosis factor-alpha on HLA-DR expression, proliferation, collagenase production and granulocyte macrophage colony-stimulating factor production by rheumatoid arthritis synoviocytes. J Clin Invest 86:1790–1798
Case JP, Lafyatis R, Remmers EF, Kumkumian GK, Wilder RL (1989) Transin/stromelysin expression in rheumatoid synovium: a transformation-associated metalloproteinase secreted by phenotypically invasive synoviocytes. Am J Pathol 135:1055–1064
Sawai T, Murakami K, Ohtani Y, Kurkinnen M, Kyogoku M, Hayashi M (1990) Stromelysin synthesizing cells in the synovial tissues of rheumatoid arthritis demonstrated by in situ hybridization and immunohistochemical methods. Tohoku J Exp Med 160:285–286
Gravallese EM, Darling JM, Ladd AL, Katz JN, Glimcher LH (1991) In situ hybridization studies of stromelysin and collagenase messenger RNA expression in rheumatoid synovium. Arthritis Rheum 34:1076–1084
Frisch SM, Clark EJ, Werb Z (1987) Coordinate regulation of stromelysin and collagenase genes determined with cDNA probes. Proc Natl Acad Sci USA 84:2600–2604
Bromley M, Woolley DE (1984) Histopathology of the rheumatoid lesion. Identification of cell types at sites of cartilage erosion. Arthritis Rheum 27:857–863
Suzuki K, Enghild JJ, Morodomi T, Salvesen G, Nagase H (1990) Mechanisms of activation of tissue procollagenase by matrix metalloproteinase 3 (stromelysin). Biochemistry 29:10261–10270
Welgus HG, Campbell EJ, Cury JD, Eisen AZ, Senior RM, Wilhelm SM, Goldbery GI (1990) Neutral metalloproteinases produced by human mononuclear phagocytes. Enzyme profile, regulation and expression during cellular development. J Clin Invest 86:1496–1502
Chu CQ, Field M, Allard S, Abney E, Feldmann M, Maini RN (1992) Detection of cytokines at the cartilage/pannus junction in patients with rheumatoid arthritis: implications for the role of cytokines in cartilage destruction and repair. Br J Rheumatol 31:653–661
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tetlow, L.C., Lees, M., Ogata, Y. et al. Differential expression of gelatinase B (MMP-9) and stromelysin-1 (MMP-3) by rheumatoid synovial cells in vitro and in vivo. Rheumatol Int 13, 53–59 (1993). https://doi.org/10.1007/BF00307734
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00307734