Summary
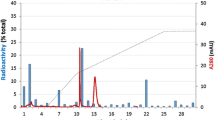
The antibody-binding ability of the glucagon-like substance in rat submaxillary gland acid saline extract was examined by affinity chromatography, and the biological activity studied using the isolated liver perfusion method. We found that the glucagon-like substances in acid saline extract could not be bound to anti-glucagon antibody and that the gel-filtration peak on ultrogel AcA 54 could increase neither glucose nor cyclic AMP output from isolated perfused rat liver. Furthermore, the radioactivity peak of 125I-glucagon on Bio Gel P-6 column chromatography moved from its original position and eluted in later fractions after incubation with an acid saline extract of the submaxillary gland. In consequence, there was 125I-glucagon degrading activity in the submaxillary gland, but no glucagon-related peptide. Therefore, it is suggested that the glucagon-like substance, which has been reported in acid saline extract of the rat salivary gland, may be an artifact due to tracer degrading activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dunbar JC, Silverman H, Kirman E, Foà PP (1977) Role of the submaxillary gland and kidney in the hyperglucagonemia of eviscerated rats. In: Foà PP, Bajaj JS, Foà NL (eds) Glucagon: its role in physiology and clinical medicine. Springer, Berlin Heidelberg New York, pp 157–166
Lawrence AM, Tan S, Hojvat S, Kirsteins L (1976) Salivary gland hyperglycemie factor: an extrapancreatic source of glucagon-like material. Science 195: 70–72
Lawrence AM, Tan S, Hojvat S, Kirsteins L, Mitton J (1976) Salivary gland glucagon in man and animals. Metabolism 25 (Suppl 1): 1405–1408
Bhathena SJ, Smith SS, Voyles NR, Penhos JC, Recant L (1977) Studies on submaxillary gland immunoreactive glucagon. Biochem Biophys Res Commun 74: 1574–1585
Smith S, Mazur A, Voyles N, Bhathena S, Recant L (1977) Is submaxillary gland immunoreactive glucagon important in carbohydrate homeostasis? Metabolism 28: 343–347
Perez-Castillo A, Blazques E (1980) Synthesis and release of glucagon by human salivary glands. Diabetologia 19: 123–129
Perez-Castillo A, Blazques E (1980) Tissue distribution of glucagon, glucagon-like immunoreactivity, and insulin in rat. Am J Physiol 238: E258-E266
Hatton TW, Kovacevic N, Dutozak M, Vranic M (1982) Glucagon-like immunoreactants in extracts of the rat hypothalamus. Endocrinology 111: 572–577
Tahara Y, Shima K, Hirota H, Ikegami H, Tanaka A, Kumahara Y (1983) Salivary gland glucagon is a fictitous substance due to tracer-degrading activity resistant to protease inhibitors. Biochem Biophys Res Commun 113: 340–347
Kenny AM (1955) Extractable glucagon of the human pancreas. J Clin Endocrinol Metab 15: 1089–1105
Tominaga M, Ebitani I, Marubashi S, Kamimura T, Katagiri T, Sasaki H (1981) Species difference of glucagon-like materials in the brain. Life Sci 29: 1577–1581
Faloona GR, Unger RH (1974) Glucagon: In: Jaffe BM, Behrman HR (eds) Methods of hormone radioimmunoassay. Academic Press, New York, pp 317–330
Yanaihara N, Nishino T, Kodaira K, Imagawa T, Nishida S, Mihara S, Yanaihara C (1979) Characterization of anti-glucagon sera elicited against a C-terminal fragment of pancreatic glucagon and their use in glucagon radioimmunoassay. In: Baba S, Kaneko T, Yanaihara N (eds) Proinsulin, insulin, C-peptide. Elsevier, North-Holland Amsterdam, pp 426–431
Heding LG, Frandesn EK, Jacobsen H (1976) Structure-function relationship: immunologic. Metabolism 25 (Suppl 1): 1327–1329
Nishino T, Kodaira T, Shin S, Imagawa K, Yanaihara N, Shima K, Kumahara Y (1981) Production of antisera to des-Asn28-Thr29-Homoser27-glucagon; the development of radioimmunoassay for total glucagon-like immunoreactivity in human plasma. Endocrinol Jpn 28: 419–427
Sugano T, Suda T, Shimada M, Oshino N (1978) Biochemical and ultrastructural evaluation of isolated rat liver systems perfused with a hemoglobin-free medium. J Biochem 83: 995–1007
Kunitada S, Honma M, Ui M (1978) Increase in plasma cyclic AMP dependent on endogenous catecholamine. Eur J Pharmacol 48: 159–169
Hunter WM, Greenwood FC (1962) Preparation of iodine-131 labelled human growth hormone of high specific activity. Nature 194: 495–496
Jorgensen KH, Larson UD (1972) Purification of 125I-glucagon by anion exchange chromatography. Horm Metab Res 4: 223–224
Tager HS, Patzelt C, Associan RK, Chan SJ, Duguid JR, Steiner DF (1980) Biosynthesis of islet cell hormones. Ann NY Acad Sci 343: 133–147
Thim L, Moody AM (1981) The primary structure of porcine glycentin (proglucagon). Regul Pept 2: 139–150
Reichlin M, Schnure JJ, Vance VK (1968) Induction of antibodies to porcine ACTH in rabbits with nonsteroidogenic polymers of BSA and ACTH. Proc Soc Exp Biol Med 128: 347–350
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tominaga, M., Yamatani, K., Marubashi, S. et al. 125I-glucagon-degrading activity in acid-saline extracts of rat salivary gland. Diabetologia 27, 392–396 (1984). https://doi.org/10.1007/BF00304856
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00304856