Summary
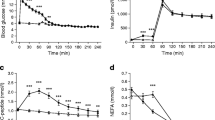
The insulin effect, evaluated with the euglycaemic clamp technique, was studied before and after hypoglycaemia in 7 patients with Type 1 (insulin-dependent) diabetes. Following an initial 2 h clamp (clamp I) hypoglycaemia was induced and 2 h later a second clamp (clamp II), identical to the former, was performed. Each subject was studied twice; during infusion with saline (placebo) or propranolol. Glucose production and disposal were studied with the 3(3H)glucose technique. During placebo infusion, hypoglycaemia elicited an insulin resistance leading to approx. 50% reduction in the steady state glucose infusion rate during clamp II as compared to clamp I (clamp I 2.58±0.32, clamp II 1.26±0.08 mg·kg−1·min−1, p<0.02). The insulin resistance was prevented by infusing propranolol (clamp I 2.29±0.29, clamp II 2.85±0.56 mg·kg−1·min−1). The posthypoglycaemic insulin resistance was due to a less pronounced insulin effect on both glucose production (clamp I 0.29±0.21, clamp II 0.86±0.19 mg·kg−1·min−1, p<0.05) and glucose utilisation (clamp I 2.84±0.26, clamp II 2.13±0.23 mg·kg−1·min−1, p<0.05). The insulin resistance on both glucose production and utilisation was prevented by propranolol. Thus, the present study demonstrates that hypoglycaemia elicits a prolonged insulin resistance which is due to a less pronounced effect of insulin to both inhibit splanchnic glucose production and to stimulate peripheral glucose utilisation. The insulin resistance is due to β-adrenergic stimulation and can be prevented by propranolol.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Somogyi M (1959) Exacerbation of diabetes by excess insulin action. Am J Med 26: 169–191
Gerich J, Davis J, Lorenzi M, Rizza R, Bohannon N, Karam J, Lewis S, Kaplan R, Schulte T, Cryer P (1979) Hormonal mechanisms of recovery from insulin-induced hypoglycaemia in man. Am J Physiol 236 (4): E380-E385
Rizza R, Cryer PE, Gerich JE (1979) Role of glucagon, catecholamines, and growth hormone in human glucose counteregulation. J Clin Invest 64: 62–71
Garber AJ, Cryer PE, Santiago JV, Haymond MW, Pagliara AS, Kipnis DM (1976) The role of adrenergic mechanisms in the substrate and hormonal response to insulin-induced hypoglycemia in man. J Clin Invest 58: 7–15
Bolli GB, Dimitriadis GD, Pehling GB, Baker BA, Morey WH, Cryer PE, Gerich JE (1984) Abnormal glucose counterregulation after subcutaneous insulin in insulin-dependent diabetes mellitus. N Engl J Med 310: 1706–1711
Gale EAM, Kurte AB, Tattersall RB (1980) In search of the Somogyi effect. Lancet 2: 279–282
Bolli GB, Gottesman IS, Campbell PJ, Haymond MW, Cryer PE, Gerich JE (1984) Glucose counterregulation and waning of insulin in the Somogyi phenomenon (posthypoglycemic hyperglycemia). N Engl J Med 311: 1214–1219
The DCCT Research Group (1984) Results f feasibility phase of the diabetes control and complications trial (DCCT): glycemic control, follow-up and complications of therapy. Diabetes 35 [Suppl 1]: 10A
Dahl-Jörgensen, K, Brichmann-Hansen O, Hanssen KF, Ganes T, Kierulf P, Smeland E, Sandvik L, Aagenaes Ö (1986) Effect of near normoglycaemia for two years on progression of early diabetic retinopathy, nephropathy, and neuropathy: the Oslo study. Br Med J 293: 1195–1199
National diabetes data group (1979) Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes 28: 1039–1057
Ewing DJ, Campbell IW, Murray A, Neilson JMM, Clarke BF (1978) Immediate heart-rate response to standing: simple test for autonomie neuropathy in diabetes. Br Med J 1: 145–147
Mackay JD, Page M McB, Cambridge J, Watkins PJ (1980) Diabetic autonomic neuropathy. Diabetologia 18: 471–478
De Fronzo RA, Tobin JD, Andres R (1979) Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol 237: E214–223
Kuzuya H, Blix PM, Horowitz DL, Steiner DF, Rubenstein AH (1979) Determination of free and total insulin and C-peptide in insulin-treated diabetics. Diabetes 26: 22–29
von Schenck H (1977) Production and characterization of an antiserum against pancreatic glucagon. Clin Chim Acta 80: 455–463
von Schenck, Nilsson U (1981) Radioimmunoassay of extracted glucagon compared with three non-extraction assays. Clin Chim Acta 109: 182–191
Eriksson BM (1981) Aluminium foil instead of glass plates for thinlayer chromatography in radioenzymatic assay. Clin Chem 27: 341–342
Holme I (1980) Determination of HbAlc with a cation exchange (mono-S) method. Academic dissertation, University of Gothenburg
DeBodo RCR, Steele N, Altszuler N, Dünn A, Bishop JS (1963) On the hormonal regulation of carbohydrate metabolism. Studies with C14-glucose. Recent Prog Horm Res 19: 445–488
Cowan JS, Hetenyi G (1971) Glucoregulatory responses in normal and diabetic dogs recorded by a new tracer method. Metabolism 20: 360–372
Radziuk J, Norwich KM, Vranic M (1978) Experimental validation of measurements of glucose turn-over in non-steady state. Am J Physiol 234: E84-E93
Perriello G, De Feo P, Calcinaro F, Ventura MM, Basta G, Torlone E, Santeusanio F, Brunetti P, Bolli GB (1987) Nocturnal hypoglycemia exaggerates fasting and postbreakfast hyperglycemia in CSII-treated IDDM. Diabetes 36: [Suppl 1] 3A
Buysschaert M, Marchand E, Ketelslegers JM, Lambert AE (1983) Comparison of plasma glucose and plasma free insulin during CSII and intensified conventional insulin therapy. Diabetes Care 6: 1–5
Bending JJ, Pickup JC, Andrea BM, Collins CG, Keen H (1985) Rarity of a marked “dawn phenomen” in diabetic subjects treated by continuous subcutaneous insulin infusion. Diabetes Care 8: 28–44
Attvall S, Eriksson B-M, Fowelin J, von Schenck H, Lager I, Smith U (1987) Early posthypoglycemic insulin resistance in man is mainly an effect of β-adrenergic stimulation. J Clin Invest 80: 437–442
Lager I, von Schenck H, Smith U (1984) Improved but not normalized glucose counter-regulation during glucagon infusion in type I (insulin-dependent) diabetes. Diabetologia 26: 337–342
Bolli G, De Feo P, Compagnucci P, Cartechini MG, Angeletti G, Santeusanio F, Brunetti P, Gerich JE (1983) Abnormal glucose counterregulation in insulin-dependent diabetes mellitus. Diabetes 32: 134–141
Lager I, Blohmé G, Smith U (1979) Effect of cardioselective and non-selective β-blockade on the hypoglycaemic response in insulin-dependent diabetics. Lancet 1: 458–462
Popp DA, Shah SD, Cryer PE (1982) Role of epinephrinemediated β-adrenergic mechanisms in hypoglycemie glucose counterregulation and posthypoglycemic hyperglycemia in insulin-dependent diabetes mellitus. J Clin Invest 69: 315–326
De Feo P, Bolli G, Perriello G, De Cosmo S, Compagnunucci P, Angeletti G, Santeusanio F, Gerich JE, Motolese M, Brunetti P (1983) The adrenergic contribution to glucose counterregulation in type I diabetes mellitus. Diabetes 32: 887–893
Gerich JE, Cryer P, Rizza R (1980) Hormonal mechanisms in acute glucose counterregulation: the relative roles of glucagon, epinephrine, norepinephrine, growth hormone, and cortisol. Metabolism 29: 1114–1118
Lager I, Jagenburg R, von Schenck H, Smith U (1980) Effect of beta-blockade on hormone release during hypoglycaemia in insulin-dependent diabetics. Acta Endocrinol 95: 364–371
Kleinbaum J, Shamoon H (1984) Effect of propranolol on delayed glucose recovery after insulin-induced hypoglycaemia in normal and diabetic subjects. Diabetes Care 7: 155–162
Popp DA, Tse TF, Shah SD, Clutter WE, Cryer PE (1984) Oral propranolol and metoprolol both impair glucose recovery from insulin-induced hypoglycemia in insulin-dependent diabetes mellitus. Diabetes Care 7: 243–247
Doberne L, Greenfield MS, Schulz B, Reaven GM (1981) Enhanced glucose utilization during prolonged glucose clamp studies. Diabetes 30: 829–835
Clutter WE, Bier DM, Shah SD, Cryer PE (1980) Epinephrine plasma metabolic clearance rates and physiological thresholds for metabolic and hemodynamic actions in man. J Clin Invest 66: 94–101
Lönnroth P, Smith U (1983) Beta-adrenergic dependent down-regulation of insulin binding in rat adipocytes. Biochem Biophys Res Commun 112: 972–979
Pessin JE, Gitomer W, Oka Y, Oppenheimer CL, Czech MP (1983) β-adrenergic regulation of insulin and epidermal growth factor receptors in rat adipocytes. J Biol Chem 258: 7386–7394
Lönnroth P, Wesslau C, Stenström G, Tisell LE, Smith U (1985) Reduced insulin binding to human fat cells following beta-adrenergic stimulation — experimental evidence and studies in patients with a phaeochromocytoma. Diabetologia 28: 901–906
Green A (1983) Catecholamines inhibit insulin-stimulated glucose transport in adipocytes in the presence of adenosine deaminase. FEBS Lett 152: 261–264
Smith U, Kuroda M, Simpson IA (1984) Counter-regulation of insulin stimulated glucose transport by catecholamines in the isolated rat adipose cell. J Biol Chem 259: 8758–8763
Kirsch DM, Baumgartner M, Rinninger T, Kemmler W, Häring HU (1983) Catecholamine-induced resistance of glucose transport in isolated rat adipocytes. Biochem J 216: 737–745
Lager I, Attvall S, Eriksson BM, von Schenck H, Smith U (1986) Studies on the insulin-antagonistic effect of catecholamines in normal man. Diabetologia 29: 409–416
Rizza RA, Mandarino LJ, Gerich JE (1982) Cortisol induced insulin resistance in man: impaired suppression of glucose production and stimulation of glucose utilization due to a postreceptor defect of insulin action. J Clin Endocrinol Metab 54: 131–138
Rizza RA, Mandarino LJ, Gerich JE (1982) Effects of growth hormone in insulin action in man. Diabetes 31: 662–669
Brattusch-Marrain PR, Smith D, De Fronzo RA (1982) The effect of growth hormone on glucose metabolism and insulin secretion in man. J Clin Endocrinol Metab 55: 973–982
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Attvall, S., Fowelin, J., von Schenck, H. et al. Insulin resistance in Type 1 (insulin-dependent) diabetes following hypoglycaemia — evidence for the importance of β-adrenergic stimulation. Diabetologia 30, 691–697 (1987). https://doi.org/10.1007/BF00296990
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00296990