Abstract
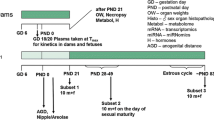
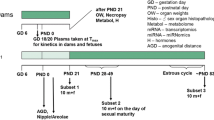
Reproduction and teratology studies were performed with etretin, the free acid analog of the retinoid etretinate. The lowest teratogenic dose of etretin in mice, rats and rabbits was 3, 15 and 0.6 mg/kg, respectively. In all three species, the lowest dose which was embryolethal, fetolethal or reduced the survivability of the pups during lactation was 2–3 times higher than the above doses. In rats, the lowest effective dose of etretin was 3 mg/kg in both the study for fertility and general reproductive performance and the peri- and postnatal study. The main adverse effect in these two experiments was a reduced survival of the F1-generation.
Similar content being viewed by others
References
Bollag W (1983) Vitamin A and retinoids: from nutrition to pharmacotherapy in dermatology and oncology. Lancet 860–863
Bollag W (1985) New retinoids with potential use in humans. In: Retinoids: newtrends in research and therapy (Saurat JH, ed), Karger, Basle, pp 274–288
Geelen JAG (1979) Hypervitaminosis A induced teratogenesis. Crit Rev Toxicol 6: 351–375
Happle R, Traupe H, Bounameaux Y, Fisch T (1984) Teratogene Wirkung von Etretinat beim Menschen. Dtsch Med Wschr 109: 1476–1480
Kistler A (1981) Teratogenesis of retinoic acid in rats: susceptible stages and suppression of retinoic acid-induced limb malformations by cycloheximide. Teratology 23: 25–31
Kochhar DM (1973) Limb development in mouse embryos. I. Analysis of teratogenic effects of retinoic acid. Teratology 7: 289–298
Lüdin E (1985) A test procedure based on ranks for the statistical evaluation of toxicological studies. Arch Toxicol 58: 57–58
Matthews LM, Johnson EM, Newman LM (1981) Introduction of the late gestational teratogenesis in rat lung by hypervitaminosis A. Teratology 23: 253–258
Newmann LM, Johnson EM, Cadogan ASA (1982) Intralitter variation of neonatal rat lung morphology following prenatal maternal hypervitaminosis A. Anat Rec 204: 153–160
Newman LM, Johnson EM, Cadogan ASA (1983) Oxygen consumption and survival prediction in neonatal rats exposed to prenatal hypervitaminosis A. Teratology 28: 219–227
Paravicini U, Busslinger A (1984) Etretinate and isotretinoin, two retinoids with different pharmacokinetic profils. In: Retinoid therapy (Cunliffe WJ, Miller AJ, eds), MTP Press Ltd., Lancaster, pp 11–23
Paravicini U, Camenzind M, Gorer M, Geiger JM, Saurat JH (1985) Multiple dose pharmacokinetics of Ro 10-1670, the main metabolite of TIGASON. In: Retinoids: new trends in research and therapy (Saurat JH, ed), Karger, Basle, pp 289–292
Peck GL (1981) Retinoids in clinical dermatology. In: Progress in diseases of the skin (Fleishmajer R, ed), vol 1, Grune and Stratton, New York, pp 227–269
Peck GL (1983) Retinoids: therapeulic use in dermatology. Curr Ther 24: 103–112
Rosa FW (1983) Teratogenicily of isotretinoin. Lancet 2: 513
Stern RS, Rosa F, Baum C (1984) Isolrelinoin and pregnancy. J Am Acad Dermalol 10: 851–854
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kistler, A., Hummler, H. Teratogenesis and reproductive safety evaluation of the retinoid etretin (Ro 10-1670). Arch Toxicol 58, 50–56 (1985). https://doi.org/10.1007/BF00292617
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00292617