Abstract
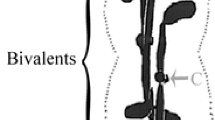
During the first meiotic division in crane-fly spermatocytes, the two homologs of a metaphase bivalent each bear two sister kinetochores oriented toward the same pole. We have previously reported treatments that increase the percentage of metaphase bivalents in which one or both homologs have bipolar malorientations: kinetochore microtubules] extending from a homolog toward both poles. The maloriented homologs lag at anaphase. Treatments that induce this behavior include: (a) recoverey from exposure to low temperatures or Colcemid or Nocodazole concentrations that prevent spindle formation but allow nuclear membrane breakdown, and (b) exposure to 6° C, a temperature that permits spindle assembly but slows progression through meiosis. Giemsa staining methods reveal two 0.5 μm diameter dots at the centromeric region of each metaphase homolog; these often are more separated in maloriented homologs. This investigation was undertaken to assess whether this separation precedes the establishment of bipolar malorientation, and hence may be a cause of it, or is only a consequence of forces resulting from bipolar malorientation. Analysis showed that, in untreated cells, the average center-to-center distance between sister centromeric dots increases during the course of meiosis I. After the above-mentioned treatments, center-to-center distances similar to those normally seen in untreated half-bivalents at anaphase I were seen in bivalents, both after and before nuclear membrane breakdown. Longer exposure to temperatures that arrested meiosis increased the degree of dot separation. Based on our data, we conclude that normal orientation during the first meiotic division is aided by the close apposition of centromeric dots, and that a time-dependent maturation occurs causing centromeric dots to separate for the second meiotic division and facilitating orientation of sister kinetochores to opposite poles. If centromeric maturation occurs either prior to or during early stages of the first meiotic division, then it may contribute to persisting bipolar malorientation.
Similar content being viewed by others
References
Ault JG, Lin H-PP (1984) Bivalent behavior in Drosophila melanogaster males containing the In (1)sc4Lsc8R X chromosome. Chromosoma 90:222–228
Bajer AS, Mole-Bajer (1972) Spindle dynamics and chromosome movements. Int Rev Cytol [Suppl] 3:40–44
Brinkley BR, Stubblefield E (1970) Ultrastructure and interaction of the kinetochore and centriole in mitosis and meiosis. Adv Cell Biol 1:119–185
Brinkley BR, Brenner SL, Hall JM, Tousson A, Balczon RD, Valdivia MM (1986) Arrangements of kinetochores in mouse cells during meiosis and spermiogenesis. Chromosoma 94:309–317
Bruning JL, Kintz BL (1977) Computational handbook of statistics. Scott, Foresman and Company, Glenview, Illinois
Calarco P (1972) The kinetochore in oocyte maturation. In: Biggers JD, Schuetz AW (eds) Oogenesis. University Park Press, Baltimore, pp 65–86
Chandley AC (1985) Maternal aging as the important etiological factor in human aneuploidy. In: Dellarco VL, Voytek PE, Hollaender A (eds) Aneuploidy. Etiology and mechanisms. Plenum Press, New York, pp 279–290
Church K, Lin H-PP (1982) Meiosis in Drosophila melanogaster. II. The prometaphase-I kinetochore micro tubule bundle and kinetochore orientation in males. J Cell Biol 93:365–373
Crozier RH (1968) An acetic acid dissociation, air-drying technique for insect chromosomes, with aceto-lacetic orcein staining. Stain Technol 43:171–173
Daniel A (1979) Single Cd band in dicentric translocations with one suppressed centromere. Hum Genet 48:85–91
Eiberg H (1974) New selective Giemsa technique for human chromosomes, Cd staining. Nature 248:55
Goldstein LSB (1981) Kinetochore structure and its role in chromosome orientation during the first meiotic division in male D. melanogaster. Cell 25:591–602
Hsu TC, Satya-Prakash KL (1985) Aneuploidy induction by mitotic arrestants in animal cell systems: possible mechanisms. In: Dellarco VL, Voytek PE, Hollaender A (eds) Aneuploidy. Etiology and mechanisms. Plenum Press, New York, pp 279–290
Janicke MA, LaFountain JR Jr (1982) Chromosome segregation in crane-fly spermatocytes: cold treatment and cold recovery induce anaphase lag. Chromosoma 85:619–631
Janicke MA, LaFountain JR Jr (1984) Malorientation of halfbivalents at anaphase: analysis of autosomal laggards in untreated, cold-treated and cold-recovering crane-fly spermatocytes. J Cell Biol 98:859–869
Janicke MA, LaFountain JR Jr (1986) Bivalent orientation and behavior in crane-fly spermatocytes recovering from cold exposure. Cell Motil Cytoskeleton 6:492–501
Kubai DF, Wise W (1981) Nonrandom chromosome segregation in Neocurtilla (Gryllotalpa) hexadactyla: An ultrastructural study. J Cell Biol 88:281–293
Ladrach KS, LaFountain JR Jr (1986) Malorientation and abnormal segregation of chromosomes during recovery from Colcemid and Nocodazole. Cell Motil Cytoskeleton 6:419–427
LaFountain JR Jr (1983) Chromosome movement during meiotic prophase in crane-fly spermatocytes. II. Analysis of polarization of chromosomes and their association with the nuclear envelope. Cell Motil 3:261–271
LaFountain JR Jr (1985a) Chromosome segregation and spindle structure in crane-fly spermatocytes following Colcemid treatment. Chromosoma 91:329–336
LaFountain JR Jr (1985b) Malorientation in half-bivalents at anaphase in crane-fly spermatocytes following Colcemid treatment. Chromosoma 91:337–346
LaFountain JR Jr (1985c) Chromosome movement during meiotic prophase in crane-fly spermatocytes: III. Microtubules and the effects of Colcemid, Nocodazole, and Vinblastine. Cell Motil 5:393–413
Lin H-PP, Church K (1982) Meiosis in Drosophila melanogaster. III. The effect of orientation disruptor (ord) on gonial mitotic and the meiotic divisions in males. Genetics 102:751–770
Lin H-PP, Ault JG, Church K (1981) Meiosis in Drosophila melanogaster. I. Chromosome identification and kinetochore microtubule numbers during the first and second meiotic division in males. Chromosoma 83:507–521
Müller W (1972) Elektronenmikroskopische Untersuchungen zum Formwechsel der Kinetochoren während der Spermatocytenteilungen von Pales ferruginea (nematocera). Chromosoma 38:139–172
Murray AW, Szostak J (1985) Chromosome segregation in mitosis and meiosis. Annu Rev Cell Biol 1:289–315
Nicklas RB (1971) Mitosis. Adv Cell Biol 2:225–297
Nicklas RB (1977) Chromosome distribution: experiments on cell hybrids and in vitro. Philos Trans R Soc Lond B 277:267–276
Nicklas RB (1985) Mitosis in eukaryotic cells: an overview of chromosome distribution. In: Dellarco VL, Voytek PE, Hollaender A (eds) Aneuploidy. Etiology and mechanisms. Plenum Press, New York, pp 183–195
Rieder CL (1982) The formation, structure, and composition of the mammalian kinetochore and kinetochore fiber. Int Rev Cytol 79:1–58
Rieder CL, Nowogrodzki R (1983) Intranuclear membranes and formation of the first meiotic spindle in Xenos peckii (Acroschismus wheeleri) oocytes. J Cell Biol 97:1144–1155
Scarcello LA, Janicke MA, LaFountain JR Jr (1986) Kinetochore microtubules in crane-fly spermatocytes: Untreated, 2° C-treated, and 6° C-grown spindles. Cell Motil Cytoskeleton 6:428–438
Schrader F (1939) The structure of the kinetochore at meiosis. Chromosoma 1:230–237
Stack SS (1974) Differential Giemsa staining of kinetochores and nucleolus organizer heterochromatin in mitotic chromosomes of higher plants. Chromosoma 47:361–378
Sumner AT (1972) A simple technique for demonstrating centromeric heterochromatin. Exp Cell Res 75:304–306
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Janicke, M.A., LaFountain, J.R. Centromeric dots in crane-fly spermatocytes: meiotic maturation and malorientation. Chromosoma 98, 358–367 (1989). https://doi.org/10.1007/BF00292389
Issue Date:
DOI: https://doi.org/10.1007/BF00292389