Summary
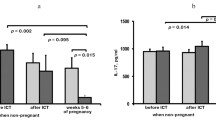
Lymphocytes bearing surface antigens indicating early and full activation have been evaluated, in addition to T cell subsets, in blood samples from diabetic pregnant patients, neonates from diabetic mothers and control groups. The type of diabetes and the trimester of pregnancy were taken into account. Monoclonal antibodies were used to enumerate total T cells, helper/inducer, cytotoxic/suppressor T lymphocytes and activated mononuclear cells using antibodies binding lymphocyte surface antigens as markers of early lymphocyte activation, and MHC Class II surface antigens as markers of late activation. A decrease in T-helper cells during the third trimester of pregnancy in Type 1 (insulin-dependent) and gestational diabetic patiens (p<0.02) and a decrease in T-suppressor cells in Type 2 (non-insulin-dependent) diabetic pregnant patients during the third trimester (p<0.01) were observed in relation to normal values. As in normal pregnancy, 4F2-positive cells were increased in 48% of diabetic pregnant patients during the second and third trimesters of gestation. Class II-positive cells were increased in almost 60% of Type 1 and gestational diabetic patients during the last trimester of pregnancy in comparison with normal pregnant women and control subjects. A decrease in T-helper cells (p<0.02) and a clear increase in 4F2-positive cells (p<0.001) and Class II-positive lymphocytes (p<0.005) were observed in the infants of diabetic mothers in comparison with control subjects. The maternal cellular immune system, actively alerted in pregnancy, is fully activated in a number of Type 1 and gestational diabetic pregnant patients. Activated lymphocytes are even found in the neonates of diabetic mothers, but these do not trigger the events leading to the onset of diabetes in the short term.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gleicher N, Siegel I (1983) Immunology of pregnancy. Wiener Klinische Wochenschrift 95, 11: 377–382
Shulman S, Dondero F, Nicotra M (eds) (1982) Immunological factors in human reproduction. Serono Symposia, vol 45. Academic Press, New York
Andreani D, Di Mario U, Federlin KF, Heding LG (1984) Immunology in diabetes. Kimpton Medical Publications, London Edinburgh
Andreani D, Di Mario U, Zuccarini O, Franco M, Sensi M, Iavicoli M, Pozzilli P (1984) Lymphocyte subsets and immune complexes in long standing diabetic patients: relation with the presence of microangiopathy. Immunol Lett 8: 17–21
Ginsberg-Fellner F, Mark EM, Nechemias C, Hausknecht RU, Rubinstein P, Dobersen MJ, Notkins AR (1980) Islet cell antibodies in gestational diabetics. Lancet 2: 362–363
Sensi M, Pozzilli P, Di Mario U (1984) A new method to facilitate the reading of up to 60 monoclonal antibody-fluorescence stained cells using medicell HLA-DR plates. J Clin Lab Immunol 13: 101–102
Eisenbarth GS, Haynes BF, Schorer JA, Fauci AS (1980) Production of monoclonal antibodies reacting with peripheral blood mononuclear cell surface differentiation antigens. J Immunol 124: 1237–1241
Lampson LA, Levy R (1980) Two populations of Ia like molecules on a human B cell line. J Immunol 125: 293–299
Guy K, Van Heyningen V, Cohen BB, Deane DL, Steel CL (1982) Differential expression and serologically distinct subpopulations of human Ia antigens detected with monoclonal antibodies to Ia alpha and beta chains. Eur J Immunol 12: 942–948
Di Mario U, Guy K (1984) Evaluation of class II antigen-positive T cells: a simple double staining method using monoclonal antibodies. J Clin Lab Immunol 13: 203–204
Tingle AJ, Lim G, Wright VJ, Dimmick JE, Hunt JA (1979) Transplacental passage of islet cell antibody in infants of diabetic mothers. Pediat Res 13: 1323–26
Steel JM, Irvine WJ, Clarke BF (1980) The significance of pancreatic islet cell antibody and abnormal glucose tolerance during pregnancy. J Clin Lab Immunol 4: 83–5
Mylvaganam R, Stowers JM, Steel JM, Wallace J, MacHendry JC, Wright AD (1983) Insulin immunogenicity in pregnancy: maternal and fetal studies. Diabetologia 24: 19–25
Fallucca F, Di Mario U, Gargiulo P, Iavicoli M, Galfo C, Contreas G, Pachi A, Andreani D (1985) Humoral immunity in diabetic pregnancy: interrelationships with maternal/neonatal complications and maternal metabolic control. Diab Metab 11: 386–395
Theophilopoulos AN, Gleicher N, Pereira AB, Dixon FJ (1981) The biology of immune complexes and their possible role in pregnancy. Am J Reprod Immunol 1: 92–105
Vazquez-Escobosa C, Perez-Medina R, Gomez-Estrada H (1983) Circulating immune complexes in hypertensive disease of pregnancy. Obst Gynecol 62: 45–48
Faulk WP, Galbraith RM, Keane M (1980) Immunological consideration of the feto-placental unit in maternal diabetes. In: Irvine WJ (ed) Immunology in diabetes. Teviot, Edinburgh, pp 309–317
Di Mario U, Fallucca F, Gargiulo P, Tiberti C, Scardellato A, Arduini P, Pachi A, Andreani D (1984) Insulin-anti-insulin complexes in diabetic women and their neonates. Diabetologia 27: 83–86
Siegel I, Gleicher N (1980) The development of the fetal immune system. Mount Sinai J Med 47: 474–480
Gill TJ (1977) Chimerism in humans. Transplant Proc 9: 1423–1431
Iavicoli M, Fallucca F, Di Mario U, Tinelli F, Trovati A, Colletti A, Montenero P, Andreani D (1980) Islet cell antibodies in the offspring of two diabetic parents. In: Pinchera A, Doniach D, Fenzi GF, Baschieri L (eds) Autoimmune aspects of endocrine disorders. Academic Press, London, pp 311–312
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Di Mario, U., Dotta, F., Gargiulo, P. et al. Immunology in diabetic pregnancy: activated T cells in diabetic mothers and neonates. Diabetologia 30, 66–71 (1987). https://doi.org/10.1007/BF00274573
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00274573