Summary
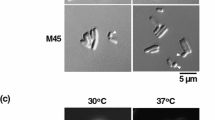
Among temperature-sensitive mutants which were defective in septum formation and formed nonseptate filaments at nonpermissive temperatures three (ts31, ts341, ts526) were identified among 434 temperature-sensitive mutants isolated at random from a mutagenized population of Bacillus subtilis 168. The results of morphological observations and characterization of these mutants showed that ts31 and ts341 were septum-initiation mutants and that ts526 was a DNA elongation mutant. The above mutations, and other mutations affecting septum initiation (div355 and tms12) were mapped by PBS1-mediated transduction on the chromosome in three separate regions as follows: pur A16-ts526-div355-cysA14; metC3-(ts31, tms12)-pyrD1-recA1; ebr-2-ts341-uvrA1-hisA1-cysB3. Our results suggest that the initiation process of septum formation requires at least four kinds of gene product. In addition, the sesult obtained with ts526 suggests an intimate connection between septum initiation and DNA replication.
Similar content being viewed by others
References
Adelberg EA, Mandel M, Chen GCC (1965) Optional conditions for mutagenesis by N-methyl-N′-nitro-N-nitrosoguanidine in Escherichia coli K-12. Biochem Biophys Res Commun 18:788–795
Bachmann BJ, Low KB, Taylor AL (1976) Recalibrated linkage map of Escherichia coli K-12. Bacteriol Rev 40:116–167
Breakefield XO, Landman OE (1973) Temperature-sensitive divisionles mutant of Bacillus subtilis defective in the initiation of septation. J Bacteriol 113:985–998
Callister H, Wake RG (1977) Completion of the replication and division cycle in temperature-sensitive DNA initiation mutants of Bacillus subtilis 168 at the non-permissive temperature. J Mol Biol 117:71–84
Carlton BC (1966) Fine-structure mapping by transformation in the tryptophan region of Bacillus subtilis. J Bacteriol 91:1795–1803
Copeland JC, Marmur J (1968) Identification of conserved genetic functions in Bacillus by use of temperature-sensitive mutants. Bacteriol Rev 32:302–312
Daneo-Moore L, Shockman GD (1977) The bacterial cell surface in growth and division. In: Poste G and Nicolson GL (eds) Cell surface review, vol 4 Elsevier/Holland Publishing Co., New York, p 597
Donachie WD, Begg KJ, Lutkenhaus JF, Salmond GPC, Martinezsalas E, Vincente M (1979) Role of the ftsA gene product in control of Escherichia coli cell division. J Bacteriol 140:388–394
Donachie WD, Jones NC, Teather RT (1973) The bacterial cell cycle. Symp Soc Gen Microbiol 23:9–44
Dubnau D, Goldthwaite C, Smith I, Marmur J (1967) Genetic mapping in Bacillus subtilis. J Mol Biol 27:163–185
Helmstetter CE, Pierucci O, Weinberger M, Holmes M, Tang MS (1979) Control of cell division in Escherichia coli. In: Gunsalus C, Sokatch JR, Ornston LN (es) The bacteria, vol 7. Academic Press, Inc., New York, San Francisco, London, p 157
Higgins ML, Shockman GD (1971) Procaryotic cell division with respect to wall and membranes. Crit Rev Microbiol 1:29–72
Hoch JA (1978) Developmental genetics at the beginning of a new era. In: Chambliss G and Vary JC (eds) Spores, vol VII. American Society for Microbiology, Washington, DC p 119
Iijima T, Diesterhaft MD, Freese E (1977) Sodium effect of growth on aspartate and genetic analysis of a Bacillus subtilis mutant with high aspartase activity. J Bacteriol 129:1440–1447
Karamata D, Gross JD (1970) Isolation and genetic analysis of temperature-sensitive mutants of B. subtilis defective in DNA synthesis. Mol Gen Genet 108:277–287
Koch AL (1977) Does the initiation of chromosome replication regulate cell division? Adv Microb Physiol 16:49–98
Lutkenhaus JF, Donachie WD (1979) Identification of the ftsA gene product. J Bacteriol 137:1088–1094
Mendelson NH (1977) Cell growth and division: a genetic viewpoint. Microbiology 5–24
Mendelson NH, Cole RM (1972) Genetic regulation of cell division initiation in Bacillus subtilis. J Bacteriol 112:994–1003
Miyakawa Y., Komano T, Maruyama Y (1980) Cell-cycle-specific inhibition by chloramphenicol of septum formation and cell division in synchronized cells of Bacillus subtilis. J Bacteriol 141:502–507
Munakata N (1977) Mapping of the genes controlling excision repair of pyrimidine photoproducts in Bacillus subtilis. Mol Gen Genet 156:49–54
Robinow CF (1949) Nuclear apparatus and cell structure of rod-shaped bacteria. In: Dubos RJ (ed) The bacterial cell. Harvard University Press, Cambridge, Massachusetts, p 355
Saito H, Miura K (1963) Preparation of transforming deoxyribonucleic acid by phenol treatment. Biochim Biophys Acta 72:619–629
Sargent MG (1975) Anucleate cell production and surface extension in a temperature-sensitive chromosome initiation mutant of Bacillus subtilis. J Bacteriol 123:1218–1234
Schaeffer P, Lonesco H, Ryte A, Balassa G (1963) La sporulation de Bacillus subtilis: etude genetique et physiologique. Colloq Int N R S (Paris) 124:553–563
Tanooka H, Sakakibara Y (1968) Radioresistant nature of the transforming activity of DNA in bacterial spores. Biochim Biophys Acta 155:130–142
Van Alstyne D, Simon MI (1971) Division mutants of Bacillus subtilis: Isolation and PBS1 transduction of division-specific markers. J Bacteriol 108:1366–1379
Author information
Authors and Affiliations
Additional information
Communicated by E. Bautz
Rights and permissions
About this article
Cite this article
Miyakawa, Y., Komano, T. Study on the cell cycle of Bacillus subtilis using temperature-sensitive mutants. Molec. Gen. Genet. 181, 207–214 (1981). https://doi.org/10.1007/BF00268428
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00268428