Summary
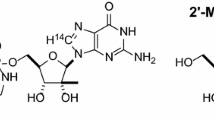
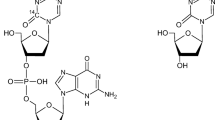
The pharmacokinetics and metabolism of dibutyl methotrexate (DBMTX) and γ-monobutyl methotrexate (γ-MBMTX) were studied in Rhesus monkeys. When a bolus IV dose of either [3H]DBMTX or [3H]γ-MBMTX was given, the principal species in serum for up to 1 h was the monoester, with MTX accounting for < 10% of the total radioactivity. Products other than γ-MBMTX and MTX were formed in substantial amounts with DBMTX, but not with γ-MBMTX. Total radioactivity recovered in the bile 5 h after [3H]DBMTX injection accounted for 32% of the administered dose, indicating high hepatic extraction for this lipophilic compound. Serum and CSF levels of unchanged γ-MBMTX, as well as of MTX arising via esterase cleavage, were measured by HPLC after IV infusion of γ-MBMTX (10 g/m2). Efflux of monoester from CSF was slower than disappearance from serum. However, γ-MBMTX levels in CSF were no higher than could be attained by infusing MTX itself at the same dose rate. While CSF/serum ratios were ca. 10-fold higher for γ-MBMTX than for MTX, this difference could be explained on the basis of the very different affinities of the two compounds for serum proteins. HPLC analysis of serum processed by methanol precipitation as opposed to ultrafiltration of the proteins showed γ-MBMTX to be >99% bound, whereas for MTX this value was 50% or less. When γ-MBMTX and MTX levels measured after ultrafiltration were corrected for this difference in serum protein binding the total amount of the two drugs in serum became almost equivalent.
Similar content being viewed by others
Abbreviations
- MTX:
-
methotrexate (4-amino-4-deoxy-N 10-methylpteroyl-l-glutamic acid)
- DBMTX:
-
dibutyl methotrexate (NSC-305985)
- γ-MBMTX:
-
γ-monobutyl methotrexate (NSC-305986)
- DDMP:
-
2,4-diamino-5-(3′,4′-dichlorophenyl)-6-methylpyrimidine (NSC-19494)
- CSF:
-
cerebrospinal fluid
- HPLC:
-
high-performance liquid chromatography
- TLC:
-
thin-layer chromatography
References
Abelson HT, Ensminger WD, Kufe D, Rosowsky A, Uren JR (1979) High-dose methotrexate-carboxypeptidase G1—A selective approach to the therapy of central nervous system tumors. In: Kisliuk RL, Brown GM (eds) Chemistry and biology of pteridines. Elsevier/North Holland, New York, pp 629–633
Beardsley GP, Rosowsky A, McCaffrey RP, Abelson HT (1979) Methotrexate di-n-butyl ester effects on thymidine metabolism. Biochem Pharmacol 28: 3069
Bourke RS, Chheda G, Bremer A, Watanabe O, Tower DB (1975) Inhibition of renal tubular transport of methotrexate by probenecid. Cancer Res 35: 110
Calvert AH, Jarman M (1979) Radiolabelled methotrexate: a warning. Lancet: 166
Calvert AH, Bondy PK, Harrap KR (1976) Some observations on the human pharmacology of methotrexate. Cancer Treat Rep 61: 1647
Chaykovky M, Rosowsky A, Modest EJ (1973) Side-chain altered methotrexate analogues designed for improved membrane transport. J Heterocycl Chem 10: 425
Chaykovsky M, Rosowsky A, Papathanasopoulos N, Chen KKN, Modest EJ, Kisliuk RL, Gaumont Y (1974) Methotrexate analogues. 3. Synthesis and biological properties of some side-chain altered analogues. J Med Chem 17: 1212
Curt GA, Tobias JS, Kramer RA, Rosowsky A, Parker LM, Tattersall MHN (1976) Inhibition of nucleic acid synthesis by the di-n-butyl ester of methotrexate. Biochem Pharmacol 25: 1943
Davidson JD, Oliverio VT (1964) The physiologic disposition of dichloromethotrexate-Cl36 in man. Clin Pharmacol 16: 321
Eisenfeld AJ, Mautner HG, Welch AD (1962) Comparative effects of amethopterin and its dimethyl ester on subcutaneous and intracerebral neoplasms. Proc Am Assoc Cancer Res 3: 316
Henderson ES, Adamson RH, Denham C, Oliverio VT (1965) The metabolic fate of tritiated methotrexate. I. Absorption, excretion, and distribution in mice, rats, dogs, and monkeys. Cancer Res 25: 1008
Jacobs SA, Stoller RG, Chabner BA, Johns DG (1977) Dose-dependent metabolism of methotrexate in man and Rhesus monkeys. Cancer Treat Rep 61: 651
Johns DG, Farquhar D, Chabner BA, Wolpert MK, Adamson RH (1973a) Antineoplastic activity of lipid-soluble dialkyl esters of methotrexate. Experientia 29: 1104
Johns DG, Farquhar D, Wolpert MK, Chabner BA, Loo TL (1973b) Dialkyl esters of methotrexate and 3′,5′-dichloromethotrexate: synthesis and interaction with aldehyde oxidase and dihydrofolate reductase. Drug Metab Dispos 1: 580
Kimelberg HK, Tracy TF, Biddlecome SF, Bourke RS (1976) The effect of entrapment in liposomes on the in vivo distribution of [3H]methotrexate in a primate. Cancer Res 36: 2649
Kimelberg HK, Biddlecome SM, Bourke RS (1977) Distribution and degradation of [3H]methotrexate after intravenous and cerebral intraventricular injection in primates. Cancer Res 37: 157
McCullough JL, Snyder DS, Weinstein GD, Friedland A, Stein B (1976) Factors affecting human percutaneous penetration of methotrexate and its analogues in vitro. J Invest Dermatol 66: 103
Piper JR, Montgomery JA, Sirotnak FM, Chello PL (1982) Synthesis of α- and γ-substituted amides, peptides, and esters of methotrexate and their evaluation as inhibitors of folate metabolism. J Med Chem 25: 182
Poplack DG, Bleyer WA, Wood HJ, Kostolick M, Savitch JL, Ommaya AK (1977) A primate model for the study of methotrexate pharmacokinetics in the central nervous system. Cancer Res 37: 1982
Rosowsky A (1973) Methotrexate analogues. 2. A facile method of preparation of lipophilic derivatives of methotrexate and 3′,5′-dichloromethotrexate by direct esterification. J Med Chem 16: 1190
Rosowsky A, Yu CS (1978) Methotrexate analogues. 10. direct coupling of methotrexate and diethyl l-glutamate in the presence of peptide bond-forming reagents. J Med Chem 21: 170
Rosowsky A, Yu CS (1979) New methods of neutral esterification of methotrexate and related compounds. In: Kisliuk RL, Brown GM (eds) Chemistry and biology of pteridines. Elsevier/North Holland, New York, pp 273–277
Rosowsky A, Ensminger WD, Lazarus H, Yu CS (1977) Methotrexate analogues. 8. Synthesis and biological evaluation of bisamide derivatives as potential prodrugs. J Med Chem 20: 925
Rosowsky A, Beardsley GP, Ensminger WD, Lazarus H, Yu CS (1978) Methotrexate analogues. 11. Unambiguous chemical synthesis and in vitro biological evaluation of α- and γ-monoesters as potential prodrugs. J Med Chem 21: 380
Rosowsky A, Lazarus H, Yuan GC, Beltz WR, Mangini L, Abelson HT, Modest EJ, Frei E III (1980) Effects of methotrexate esters and other lipophilic antifolates on methotrexate-resistant human leukemic lymphoblasts. Biochem Pharmacol 29: 648
Rosowsky A, Forsch R, Uren J, Wick M (1981) Methotrexate analogues. 14. Synthesis of new γ-substituted derivatives as dihydrofolate reductase inhibitors and potential anticancer agents. J Med Chem 24: 1450
Rubin R, Owens E, Rall DP (1968) Transport of methotrexate by the choroid plexus. Cancer Res 28: 689
Shapiro WR, Ansman JI, Rall DP (1970) Studies on the chemotherapy of experimental brain tumors: evaluation of 1,3-bis(2-chloroethyl)-1-nitrosourea, cyclophosphamide, mithramycin, and methotrexate. Cancer Res 30: 2401
Szeto DW, Cheng YC, Rosowsky A, Yu CS, Modest EJ, Piper JR, Temple C Jr, Elliott RD, Rose JD, Montgomery JA (1979) Human thymidylate synthetase. III. Structure-activity relationships. Biochem Pharmacol 28: 2633
Warren JD, Angier RB, Morton G, Rosowsky A (1980) A preparative scale synthesis and chromatographic separation of methotrexate α- and γ-monobutyl esters. Org Prep Proc Intern 12: 191–197
Watson E, Cohen JL, Chan KK (1978) High-pressure liquid chromatographic determination of methotrexate and its major metabolite, 7-hydroxymethotrexate, in human plasma. Cancer Treat Rep 62: 381
Weinstein GD, McCullough JL (1975) Effects of methotrexate esters on normal and psoriatic skin. Arch Dermatol 111: 471
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rosowsky, A., Abelson, H.T., Beardsley, G.P. et al. Pharmacologic studies on the dibutyl and γ-monobutyl esters of methotrexate in the rhesus monkey. Cancer Chemother. Pharmacol. 10, 55–61 (1982). https://doi.org/10.1007/BF00257240
Issue Date:
DOI: https://doi.org/10.1007/BF00257240