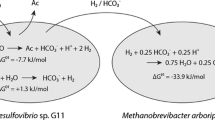
Abstract
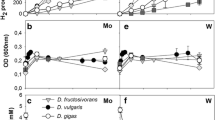
Competition for ethanol between the sulfate-reducing bacteria Desulfobulbus propionicus, Desulfotomaculum orientis, Desulfovibrio vulgaris Marburg, Desulfovibrio gigas, Desulfovibrio desulfuricans Essex and the fermenting bacteria Pelobacter propionicus and Acetobacterium carbinolicum were studied in batch culture. A number of these bacteria was also chosen for competition experiments under ethanol limitation in chemostat culture. The maximum growth rates determined by washout experiments were higher for the fermenting bacteria (μmax=0.096 resp. 0.335h−1) than for the sulfate-reducing bacteria (μmax≈0.03h−1). In contrast, the saturation concentrations for half maximum growth rates (Ks values) for ethanol were lower for the sulfate-reducing bacteria (Ks≈5 μM) than for the fermenting bacteria (Ks≥50 μM). In batch culture competition experiments the fermenting bacteria turned out to be the better competitors, degrading 51–80% of the ethanol added. In competition experiments with ethanollimited chemostat cultures the sulfate-reducing bacteria Desulfobulbus propionicus and Desulfovibrio vulgaris outcompeted Pelobacter propionicus at dilution rates below their maximum specific growth rates. At a high dilution rate, a fast growing population of Desulfobulbus propionicus originated and was enriched in the chemostat during the competition experiment.
Similar content being viewed by others
References
Bryant MP (1976) The microbiology of anaerobic degradation and methanogenesis with special reference to sewage. In: Schlegel HG, Barnea J (eds) Microbial energy conversion. Goltze, Göttingen, pp 107–117
Button DK (1985) Kinetics of nutrient-limited transport and microbial growth. Microbiol Rev 49: 270–297
Cline JD (1969) Spectrophotometric determination of hydrogen sulfide in natural waters. Limnol Oceanogr 14: 454–458
Cord-Ruwisch R, Seitz H-J, Conrad R (1987) The capacity of hydrogenotrophic bacteria to compete, for traces of hydrogen depends on the redox potential of the terminal electron accepto.. Arch Microbiol 149: 350–357
Cypionka K (1986) Sulfide-controlled continuous culture of sulfatereducing bacteria. J Microbiol Methods 5: 1–9
Dean ACR (1972) Influence of environment on the control of enzyme synthesis. J Appl Chem Biotechnol 22: 245–259
Dykhuizen DE, Hartl DL (1983) Selections in chemostats. Microbiol Rev 47: 150–181
Harder W, Kuenen JG, Matin A (1977) A Review: Microbial selection in continuous culture. J Appl Bacteriol 43: 1–24
Healy FP (1980) Slope of the Monod equation as an indicator of advantage in nutrient competition. Microb Ecol 5: 281–286
Herbert D (1958) Some principles of continuous culture. In: Tunevall G (ed) Recent progress in microbiology, 7th Int Congr Microbiol. Almquist & Wiksell, Stockholm, pp 381–396
Jannasch HW (1968) Growth characteristics of heterotrophic bacteria in seawater. J Bacteriol 95: 722–723
Kaspar HF, Wuhrmann K (1978) Product inhibition in sludge digestion. Microb Ecol 4: 241–248
Kreikenbohm R (1984) Untersuchung von definierten Mischkulturen anaerober Bakterien in kontinuierlicher Kultur. Thesis, Univ Konstanz
Kreikenbohm R, Pfennig N (1985) Anaerobic degradation of 3,4,5-trimethoxybenzoate by defined mixed cultures of Acetobacterium woodii, Pelobacter acidigallici, and Desulfobacter postgatei. FEMS Microb Ecol 31: 29–38
Kristjansson JK, Schönheit P, Thauer RK (1982) Different Ks values for hydrogen of methanogenic bacteria and sulfate reducing bacteria: an explanation for the apparent inhibition of methanogenesis by sulfate. Arch Microbiol 131: 278–282
Kuenen JG, Harder W (1982) Microbial competition in continuous culture. In: Burns RG, Slater JH (eds) Experimental microbial ccology. Blackwell, Oxford London, pp 342–367
Laanbroek HJ, Geerligs HJ, Peijnenburg AACM, Siesling J (1983) Competition, for l-lactate between Desulfovibrio, Veillonella, and Acetobacterium species isolated from anaerobic intertidal sediments. Microb Ecol 9: 341–354
Laanbroek HJ, Veldkamp H (1982) Microbial interactions in sediment communities. Philos Trans R Soc Lond B 297: 533–550
Lovley DR, Klug MJ (1982) Intermediary metabolism of organic matter in the sediment of an eutrophic lake. Appl Environ Microbiol 43: 552–560
Lovley DR, Klug MJ (1983) Sulfate reducers can outcompete methanogens at freshwater sulfate concentrations. Appl Environ Microbiol 45: 187–192
Marr AG, Nilson EH, Clark DJ (1963) The maintenance of Escherichia coli. Ann N Y Acad Sci 102: 536–548
Monod J (1942) Recherches sur la croissance des cultures bactériennes. Hermann, Paris
Neijssel OM, Tempest DW (1976) Bioenergetic aspects of aerobic growth of Klebsiella aerogenes NTCC 418 in carbon-limited and carbon-sufficient chemostat cultures. Arch Microbiol 107: 215–221
Novick A (1961) Bacteria with high level of specific enzymes. In: Growth in living systems. Purdue Growth Symposium. Basic Books, New York, pp 93–106
Pfennig N, Wagener S (1986) An improved method of preparing wet mounts for photographs of microorganisms. J Microbiol Methods 4: 303–306
Pirt SJ (1957) The oxygen requirement of growing cultures of an Aerobacter species determined by means of the continuous culture technique. J Gen Microbiol 16: 59–75
Pirt SJ (1965) The maintenance energy of bacteria in growing cultures. Proc Roy Soc B 163: 224–231
Pirt SJ (1975) Principles of microbe and cell cultivation. Blackwell, Oxford
Robinson JA, Tiedje JM (1984) Competition between sulfate-reducing and methanogenic bacteria for H2 under resting and growing conditions. Arch Microbiol 137: 26–32
Schink B (1984) Fermentation of 2,3-butanediol by Pelobacter carbinolicus sp. nov. and Pelobacter propionicus sp. nov., and evidence for propionate formation from C2 compounds. Arch Microbiol 137: 33–41
Schink B, Pfennig N (1982) Propionigenium modestum gen. nov. sp. nov., a new strictly anaerobic, nonsporing bacterium growing on succinate. Arch Microbiol 133: 209–216
Schink B, Phelps TJ, Eichler B, Zeikus JG (1985) Comparison of ethanol degrading pathways in anoxic freshwater environments. J Gen Microbiol 131: 651–660
Schönheit P, Kristjansson JK, Thauer RK (1982) Kinetic mechanism for the ability of sulfate reducers to outcompete methanogens for acetate. Arch Microbiol 132: 285–288
Tschech A, Pfennig N (1984) Growth yield increase linked to coffeate reduction in Acetobacterium woodii. Arch Microbiol 137: 163–167
Widdel F (1988) Microbiology and ecology of sulfate-and sulfurreducing bacteria. In: Zehnder ABJ (ed) Biology of anaerobic microorganisms. Wiley, New York, pp 469–585
Widdel F, Pfennig N (1981) Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids. I. Isolation of new sulfate-reducing bacteria enriched with acetate from saline environments. Appl Environ Microbiol 33: 275–281
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Szewzyk, R., Pfennig, N. Competition for ethanol between sulfate-reducing and fermenting bacteria. Arch. Microbiol. 153, 470–477 (1990). https://doi.org/10.1007/BF00248429
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00248429