Summary
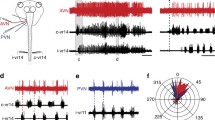
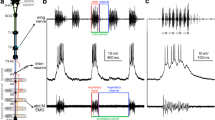
Extracellular stimulation over the dorsal funiculus in the spinal cord of lampreys was found to selectively activate prolonged episodes of fictive arousal respiration (Figs. 1, 3). The induced episodes showed comparable increases in cycle frequency and motoneuron burst duration to the spontaneous arousal pattern observed in isolated brain preparations (Fig. 2). Intracellular stimulation of primary sensory neurons with axons in the dorsal funiculus, called ‘dorsal cells’, also elicited the arousal pattern (Fig. 4). Mechanoreceptive dorsal cells respond to cutaneous stimulation. When mechanical stimuli were applied to the skin of intact lampreys (Fig. 6) or to lampreys with ipsilateral vagotomy, arousal respiration was induced (Figs. 7, 8). Bilateral, but not unilateral, trigeminal lesion blocked dorsal cell induction of the arousal response (Fig. 5). Spontaneous arousal respiration was recorded from intact, unrestrained lampreys (Fig. 9). These results suggest that fictive arousal respiration is the in vitro correlate of natural arousal respiration in lampreys, and that one mechanism leading to arousal respiration may be the activity of sensory dorsal cells. A model for respiratory motor pattern switching in lamprey is proposed. The model suggests that the normal and arousal patterns are produced by separately engaging rostral or caudal pattern generators in the medulla, rather than by modifying one pattern generator (Fig. 10).
Similar content being viewed by others
References
Buchanan JT, Brodin L, Hokfelt T, van Dongen PA, Grillner S (1987) Survey of neuropeptide-like immunoreactivity in the lamprey spinal cord. Brain Res 408:299–302
Calabrese RL, Arbas EA (1985) Modulation of central and peripheral rhythmicity in the heartbeat system of the leech. In: Selverston AI (ed) Model neural networks and behavior. Plenum, New York, pp 69–85
Claridge PN, Potter IC (1975) Oxygen consumption, ventilatory frequency and heart rate of lampreys (Lampetra fluviatilis) during their spawning run. J Exp Biol 63:193–206
Freud S (1877) Über den Ursprung der hinteren Nervenwurzeln im Rückenmark von Amocoetes (Petromyzon planeri). SB Acad Wiss Wien 75:15–30
Freud S (1878) Über Spinalganglien und Rückenmark des Petromyzon. SB Akad Wiss Wien 78:81–167
Hughes A (1957) The development of the primary sensory system in Xenopus laevis (Daudin). J Anat 91:323–328
Johansen K, Lenfant C, Hanson D (1973) Gas exchange in the lamprey Entosphenus tridentatus. Comp Biochem Physiol 44A:107–119
Johnels AG (1958) On the dorsal ganglion cells of the spinal cord in lampreys. Acta Zool Stockholm 39:201–216
Kawasaki R (1979) Breathing rhythm-generation in the adult lamprey, Entosphenus japonicus. Jpn J Physiol 29:327–338
Kawasaki R (1984) Breathing rhythm-generation mechanism in the adult lamprey (Lampetra japonica). Jpn J Physiol 34:319–335
Marder E, Hooper SL (1985) Neurotransmitter modulation of the stomatogastric ganglion of decapod crustaceans. In: Selverston AI (ed) Model neural networks and behavior. Plenum, New York, pp 319–337
Martin AR, Wickelgren WO (1971) Sensory cells in the spinal cord of the sea lamprey. J Physiol 212:65–83
Nagy F, Dickinson PS (1983) Control of a central pattern generator by an identified modulatory interneurone in Crustacea. I. Modulation of the pyloric motor output. J Exp Biol 105:33–58
Nieuwenhuys R (1977) The brain of the lamprey in a comparative perspective. Ann NY Acad Sci 299:97–145
Rovainen CM (1967) Physiological and anatomical studies on large neurons of central nervous system of the sea lamprey (Petromyzon marinus). II. Dorsal cells and giant interneurons. J Neurophysiol 30:1024–1042
Rovainen CM (1974) Respiratory motoneurons in lampreys. J Comp Physiol 94:57–68
Rovainen CM (1977) Neural control of ventilation in the lamprey. Fed Proc 36:2386–2389
Rovainen CM (1979) Neurobiology of lampreys. Physiol Rev 59:1007–1077
Rovainen CM (1983) Generation of respiratory activity by the lamprey brain exposed to picrotoxin and strychnine and weak synaptic inhibition in motoneurons. J Neurosci 10:875–882
Rovainen CM (1985) Respiratory bursts at the midline of the rostral medulla of the lamprey. J Comp Physiol A 157:303–309
Russell DF, Rovainen CM, Thompson KJ (1985) Arousal breathing in lampreys: modulation of the respiratory pattern generator. Soc Neurosci Abstr 11:477
Sigvardt KA, Grillner S, Wallén P, van Dongen PAM (1985) Activation of NMDA receptors elicits fictive locomotion and bistable membrane properties in the lamprey spinal cord. Brain Res 336:390–395
Sombati S, Hoyle G (1984) Generation of specific behaviors in a locust by local release into neuropil of the natural neuromodulator octopamine. J Neurobiol 15:481–506
Sterba G, Naumann W (1970) Neural and glial secretion in the spinal cord of the lamprey (Lampetra planeri). Acta Zool Stockholm 51:159–168
Strumwasser FL, Kaczmarek LK, Chiu AY, Heller E, Jennings KR, Viele DP (1980) Peptides controlling behavior in Aplysia. In: Bloom FE (ed) Peptides: Integrators of cell and tissue functions. Raven Press, New York, pp 197–218
Thompson KJ (1985) Organization of inputs to motoneurons during fictive respiration in the isolated lamprey brain. J Comp Physiol A 157:291–302
Wallén P, Grillner S (1987) N-methyl-D-asparate receptor-induced inherent oscillatory activity in neurons during fictive locomotion in the lamprey. J Neurosci 7:2745–2755
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thompson, K.J. Control of respiratory motor pattern by sensory neurons in spinal cord of lamprey. J Comp Physiol A 166, 675–684 (1990). https://doi.org/10.1007/BF00240017
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00240017