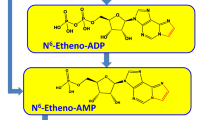
Abstract
3′-O-(4-benzoyl)benzoyl ATP (BzATP) was used as a photoaffinity analog of ATP to label potential ATP receptors in ciliated cells. Like ATP, without photoactivation, BzATP stimulated the ciliary beat frequency in tissue culture up to threefold. Irradiation of intact cells in the presence of [α-32P]BzATP followed by SDS-PAGE and autoradiography revealed two labeled proteins with molecular masses of 46 and 96 kDa (p46 and p96). Photolabeling of both proteins was susceptible to digestion with trypsin, implying that the labeled proteins are at least partially exposed on the extracellular surface of the plasma membrane. The dependence of 32P incorporation in both proteins on [α-32P]BzATP concentration was similar. Labeling of p46 but not p96 required Ca2+ or Mg2+. Various nucleotides stimulated the ciliary frequency, and inhibited the photolabeling of p46 and p96. The rank order of apparent affinity for p46 is: ATP ÃDP>GTPγS>ADP βS, UTP, 2MeSATP, AMP-PNP >AMP-PCP>AMP>adenosine; for p96 it is: ADP≅ADP β S ⩾ ATP ≫ AMP-PCP, AMP-PNP>GTPγS⩾ AMP>2MeSATP, UTP, adenosine. The rank of stimulation of ciliary beat frequency is: ADPβS, UTP⩾ 2MeSATP, GTPγS, AMP-PNP, ATP⩾ADP>AMPPCP>adenosine>AMP. These results suggest the involvement of p46 in the stimulatory effect of extracellular ATP on the ciliary beat, as a P2 purinoceptor. On the other hand, p96 may represent a P2 purinoceptor or an ectonucleotidase.
Similar content being viewed by others
References
Boyer, J.L., Cooper, C.L., Harden, T.K. 1990. [32P]3′-O-(4-benzoyl)benzoyl ATP as a photoaffinity label for a phospholipase C-coupled P2y-purinergic receptor. J. Biol. Chem. 265:13515–13520
Boyer, J.L., Harden, T.K. 1989. Irreversible activation of phospholipase C-coupled P2y-purinergic receptor by [32P]3′-O-(4-benzoyl)benzoyl ATP. Mol. Pharmacol. 36:831–835
Burnstock, G. 1978. In: Cell Membrane Receptors for Drugs and Hormones: A Multidisciplinary Approach, R.W. Straub, and L. Bolis, editors, pp. 107–118. Raven Press, New York
Burnstock, G., Buckley, N.J. 1985. The classification of receptors for adenosine and adenine nucleotides. Methods Pharmacol. 6:193–212
Burnstock, G., Kennedy, C. 1985. Is there a basis for distinguishing two types of P2-purinoceptor? Gen. Pharmacol. 16:433–440
Cusack, N.J. 1993. P2 receptors: subclassification and structureactivity relationship. Drug Dev. Res. 28:244–252
Dubyak, G.R. 1991. Signal transduction by P2-purinergic receptors for extracellular ATP. Am. J. Respir. Cell Mol. Biol. 4:295–300
Dubyak, G.R., El-Moatassim, C. 1993. Signal transduction via P2-purinergic receptors for extracellular ATP and other nucleotides. Am. J. Physiol. 265:C577-C606
Ehrlich, Y.H., Hogan, M.V., Pawlowska, Z., Naik, U., Kornecki, E. 1990. Ectoprotein kinase in the regulation of cellular responsiveness to extracellular ATP. Ann. N.Y. Acad. Sci. 603:401–416
El-Moatassim, C., Dornand, J., Mani, J.C. 1992. Extracellular ATP and cell signalling. Biochim. Biophys. Acta 1134:31–45
El-Moatassim, C., Dubyak, G.R. 1993. Dissociation of the poreforming and phospholipase D activities stimulated via P2z purinergic receptors in BAC1.2F5 macrophages. Product inhibition of phospholipase D enzyme activity. J. Biol. Chem. 268:15571–15578
Erb, L., Lustig, K.D., Ahmed, A.H., Gonzalez, F.A., Weisman, G.A. 1990. Covalent incorporation of 3′-O-(4-benzoyl)benzoylATP into a P2 purinoceptor in transformed mouse fibroblasts. J. Biol. Chem. 265:7424–7431
Erb, L., Lusting, K., Sullivan, D. Turner, J., Weisman, G. 1993. Functional expression and photoaffinity labeling of a cloned P2u purinergic receptor. Proc. Nat. Acad. Sci. USA 90:10449–10453
Eshel, D., Grossman, Y., Priel, Z. 1985. Spectral characterization of ciliary beating: Variation of frequency with time. Am. J. Physiol. 249:C160-C165
Eshel, D., Priel, Z. 1987. Characterization of metachronal beating of cilia on frogs palate epithelium in tissue culture. J. Physiol. 388:1–8
Etcheberrigaray, R., Fiedler, J.L., Pollard, H.B., Rojas, E. 1991. Endoplasmic reticulum as a source of Ca2+ in neurotransmitter secretion. Ann. N.Y. Acad. Sci. 635:90–99
Gheber, L., Priel, Z. 1989. Synchronization between beating cilia. Biophys. J., 55:183–191
Gheber, L., Priel, Z. 1990. Ciliary activity under normal conditions and under viscous load. Biorheol. 27:547–557
Gheber, L., Priel, Z. 1994. Metachronal activity of cultured mucociliary epithelium under normal and stimulated conditions. Cell Motil. Cytoskeleton 28:333–345
Gomperts, B.D. 1983. Involvement of guanine nucleotide-binding protein in the gating of Ca2+ by receptors. Nature 306:64–66
Gonzalez, F.A., Wang, D.J., Huang, N.N., Heppel, L.A. 1990. Activation of early events of the mitogenic response by a P2Y purinoceptor with covalently bound 3′-O-(4-benzoyl)benzoyladenosine 5′-triphosphate. Proc. Natl. Acad. Sci. USA 87:9717–9721
Gordon, J.L. 1986. Extracellular ATP: effects, sources and fates. Biochem. J. 233:309–319
Hargrave, P.A., McDowell, J.H. 1992. Rhodopsin and phototransduction: a model system for G protein-linked receptors. FASEB J. 6:2323–2331
James, S., Richardson, P.J. 1993. Production of adenosine from extracellular ATP at the striatal cholinergic synapse. J. Neurochem. 60:219–27
Kim, K., Zhang, Q., Wilson, A., Lee, B., Berman, J. 1994. Binding kinetics of ATPγ35S on cultured primary tracheal surface epithelial cells. Am. J. Respir. Cell Mol. Biol. 10:154–159
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lin, S.-H., Culic, O., Flanagan, D., Hixon, D.C. 1991. Immunochemical characterization of two isoforms of rat liver ecto-ATPase that show an immunological and structural identity with a glycoprotein Cell-adhesion molecule with Mr 105000. Biochem. J. 278:155–161
Lin, S.-H., Guidotti, G. 1989. Cloning and expression of a cDNA coding for a rat liver plasma membrane ecto-ATPase. J. Biol. Chem. 264:14408–14414
Mahmood, R., Yout, R.G. 1984. Photochemical probes of the active site of myosin. J. Biol. Chem. 259:1256–1259
Majid, M.A., Okajima, F., Kondo, Y. 1993. UTP activates phospholipase C-Ca2+ system through a receptor different from the 53-kDa ATP receptor in PC12 cells. Biochem. Biophys. Res. Commun. 195:415–421
Motte, S., Pirotton, S., Boeynaems, J.M. 1993. Heterogeneity of ATP receptors in aortic epithelial cells. Involvement of P2y and P2u receptors in inositol phosphate response. Circ. Res. 72:504–510
O'Connor, S.E., Dainty, I.A., Leff, P. 1991. Further subclassification of ATP receptors based on agonist studies. Trends Pharmacol. Sci. 12:137–141
Ovadiahu, D., Eshel, D., Priel, Z. 1988. Intensification of ciliary motility by extracellular ATP. Biorheol. 25:489–501
Parr, C., Sullivan, D., Paradiso, A., Lazarowski, E., Burch, L., Olsen, J., Erb, L., Weisman, G., Boucher, R., Turner, J. 1994. Cloning and expression of a human P2u nucleotide receptor, a target for cystic fibrosis pharmacology. Proc. Natl. Acad. Sci. USA 91:3275–3279
Pearson, J.D. 1985. Ectonucleotidases: Measurement of activities and use of inhibitors. Methods Pharmacol. 6:83–107
Priel, Z. 1987. Direct measurements of velocity of metachronal wave in beating cilia. Biorheol. 24:599–603
Redegeld, F., Filippini, A., Sitkovsky, M. 1991. Comparative studies of the cytotoxic T lymphocyte-mediated and cytotoxicity of extracellular ATP-induced cell lysis. Different requirements in extracellular Mg2+ and pH. J. Immunol. 147:3638–3645
Rhoads, A.R., Parui, R., Vu, N.D., Cadogan, R., Wagner, P.D. 1993. ATP-induced secretion in PC12 cells and photoaffinity labeling of receptors. J. Neurochem. 61:1657–1666
Ritov, V.B., Murzakhmetova, M.K., Tverdislova, I.L., Menshikova, E.V., Butylin, A.A., Avakian, T., Yakovenko, L.V. 1993. Alamethicin as a permeabilizing agent for measurements of Ca2+dependent ATPase activity in proteoliposomes, sealed membrane vesicles, and whole cells. Biochim. Biophys. Acta 1148:257–62
Saano, V., Virta, P., Joki, S., Nuutinen, J., Karttunen, P., Silvasti, M. 1992. ATP induces respiratory ciliostimulation in rat and guinea pig in vitro and in vivo. Rhinology 30:33–30
Soderquist, A.M., Carpenter, G. 1986. Biosynthesis and metabolic degradation of receptors for epidermal growth factor. J. Membrane Biol. 90:97–105
Tarasiuk, A., Bar-Shimon, M., Gheber, L., Korngreen, A.R.G., Priel, Z. 1995. Extracellular ATP induces hyperpolarization and motility stimulation of ciliary cells. Biophys. J. 68:1163–1169
van Rhee, A.M., van Winden, B.C., Nagelkerke, J.F., de Bont, H.J., Ijzerman, A.P., Soudijn, W. 1993. Binding of the radioligand [35S]adenosine 5′-O-(2-thiodiphosphate) and intracellular calcium response in rat liver parenchymal cells. Biochem. Pharmacol. 45:801–807
Varhaus, E.F., Deyrup, I.J. 1953. The effect of ATP on the cilia of the pharyngeal mucus of the frog. Science 118:553–554
Villalon, M., Hinds, T.R., Verdugo, P. 1989. Stimulus-response coupling in mammalian ciliated cells. Demonstration of two mechanisms of control for cytosolic [Ca2+]. Biophys. J. 56:1255–1258
Weiss, T., Gheber, L., Shoshan-Barmatz, V., Priel, Z. 1992. A possible mechanism of ciliary stimulation by extracellular ATP: involvement of Calcium dependent potassium channels and exogenous Ca2+. J. Membrane Biol. 127:185–193
Wilkinson, G., McKechnie, K., Dainty, I., Boarder, M. 1994. P2y purinoceptor and nucleotide receptor-induced relaxation of precontracted bovine aortic collateral artery rings: differential sensitivity to suramin and indomethacin. J. Pharmacol. Exp. Ther. 268:881–887
Williams, N., Ackerman, S.H., Coleman, P.S. 1986. Benzophenone-ATP: a photoaffinity label for the active site of ATPases. Methods Enzymol. 126:667–682
Williams, N.F., Coleman, P.S. 1982. Exploring the adenine nucleotide binding sites on mitochondrial F1-ATPase with a new photoaffinity probe, 3′-O-(4-benzoyl)benzoyl adenosine 5′triphosphate. J. Biol. Chem. 257:2834–2841
Wong, L.B., Yeates, D.B. 1992. Luminal purinergic regulatory mechanisms of tracheal ciliary beat frequency. Am. J. Respir. Cell Mol. Biol. 7:447–454
Zarka, A., Shoshan-Barmatz, V. 1993. Characterization and photoaffinity labeling of the ATP binding site of the ryanodine receptor from skeletal muscle. Eur. J. Biochem. 213:147–154
Zimmerman, H. 1992. 5′ nucleotidase: Molecular Structure and Functional Aspects. Biochem. J. 285:345–365
Author information
Authors and Affiliations
Additional information
This work was supported by grants (to Z.P. and to V.S-B.) from the Fund for Basic Research administered by the Israel Academy of Science and Humanities.
Rights and permissions
About this article
Cite this article
Gheber, L., Priel, Z., Aflalo, C. et al. Extracellular ATP binding proteins as potential receptors in mucociliary epithelium: Characterization using [32P]3′-O-(4-benzoyl)benzoyl ATP, a photoaffinity label. J. Membarin Biol. 147, 83–93 (1995). https://doi.org/10.1007/BF00235399
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00235399