Abstract
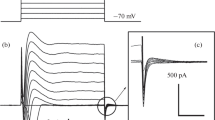
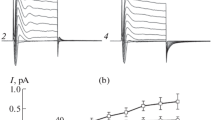
During volume regulation in hypotonic media, glial cells release a large portion of their amino acids. These amino acid losses appear to be mediated by a diffusion type of transport and a swelling-activated chloride channel seems to be involved. The objective of this project was to provide direct evidence that amino acids could diffuse through a Cl− channel. Using a human glial cell line, Cl− currents activated in hypotonic media were measured in whole-cell patch clamp. To measure the currents produced by amino acids, it was necessary to increase the pH of external solutions to basic values reaching 9.6 and 10.0 to raise the concentration of the anionic form of these amino acids. Introducing external hypotonic media containing high concentrations of amino acids, like glycine, taurine, glutamine and glutamate, it was possible to measure their respective current-voltage curves with NMDG-Cl-filled pipettes. From the reversal potentials, their permeability ratios with respect to chloride were determined. It was found that the low molecular weight amino acids, like glycine, were most permeant, while the larger ones, like glutamine, had a lower permeability with respect to chloride. The amino acids with two carboxyl groups, like glutamate, had a much lower permeability ratio. The reversal potentials for some metabolites, like lactate and malate were also measured for comparison. These results demonstrate that amino acids can diffuse through anion channels and that activation of these channels in pathological conditions could be at least partly responsible for the observed increase in external amino acids.
Similar content being viewed by others
References
Banderali, U., Roy, G. 1992. Anion channels for amino acids in MDCK cells. Am. J. Physiol. 263:C1200-C1207
Chan, H.C., Fu, W.O., Chung, Y.W., Huang, S.J., Zhou, T.S., Wong, P.Y.D. 1993. Characterization of a swelling-induced chloride conductance in cultured rat epididymal cells. Am. J. Physiol. Cell Physiol. 265:C997-C1005
Halm, D.R., Frizzell, R.A. 1992. Anion permeation in an apical membrane chloride channel of a secretory epithelial cell. J. Gen. Physiol. 99:339–366
Haynes, J.K., Goldstein, L. 1993. Volume-regulatory amino-acid transport in erythrocytes of the little skate, Raja erinacea. Am. J. Physiol. 265:R173-R179
Jackson, P.S., Morris, R., Strange, K. 1994. The volume sensitive organic osmolyte-anion channel VSOAC is regulated by nonhydrolytic ATP binding. Am. J. Physiol. 267:C1203-C1209
Jackson, P.S., Strange, K. 1993. Volume sensitive anion channels mediate swelling-activated inositol and taurine efflux. Am. J. Physiol. 265:C1489-C1500
Kimelberg, H.K., Ransom, B.R. 1986. Physiological and pathological aspects of astrocytic swelling. In: Astrocytes, Vol. 3, S. Federoff and A. Vanakis, editors, pp. 129–166. Academic Press. Orlando, F
Kimelberg, H.K., Goderie, S.K., Higman, S., Pang, S., Waniewski, R.A. 1990a. Swelling-induced release of glutamate, aspartate and taurine from astrocytes cultures. J. Neurosci. 10:1583–1591
Kimelberg, H.K., Anderson, E., Kettenmann, H. 1990b. Swellinginduced changes in electrophysiological properties of cultured astrocytes and olygodendrocytes.II. Whole-cell currents. Brain Res. 529:262–268
Kirk, K., Ellory, J.C., Young, J.D. 1992. Transport of organic substrates via a volume-activated channel. J. Biol. Chem. 267:23475–23478
Kubo, M., Okada, Y. 1992. Volume regulatory Cl channel currents in cultured human epithelial cells. J. Physiol. 456:351–371
Nilius, B., Sehrer, J., Droogmans, G. 1994. Permeation properties and modulation of volume-activated Cl currents in human endothelial cells. Brit. J. Pharmacol. 112:1049–1056
Pasantes-Morales, P., Alvez, S., Sanchez-Olea, R., Moran, J. 1993. Contribution of organic and inorganic osmolytes to volume regulation in rat brain cell in culture. Neurochem. Res. 18:445–452
Pasantes-Morales, H., Murray, R.A., Sanchez-Olea, R., Moran, J. 1994. Regulatory volume decrease in cultured astrocytes II. Permeability pathway to amino acids and polyols. Am. J. Physiol. 266:C172-C178
Ross, P.E., Garber, S.S., Cahalan, M.D. 1994. Membrane chloride conductance and capacitance in jurkat T lymphocytes during osmotic swelling. Biophys. J. 66:169–178
Roy, G., Malo, C. 1992. Activation of amino acid diffusion by a volume increase in cultured kidney (MDCK) cells. J. Membrane Biol 130:83–90
Sanchez-Olea, R., Moran, J., Martinez, A., Pesantes-Morales, H. 1991. Hypoosmolarity-activated fluxes of taurine in astrocytes are mediated by diffusion. Neurosci. Lett. 130:233–236
Stoddard, J.S., Steinbach, J.H., Simchowitz, L. 1993. Whole cell Cl− currents in human neutrophils induced by cell swelling. Am. J. Physiol. Cell Physiol. 265:C156-C165
Waltz, W., Klimaszewski, A., Paterson, I.A. 1993. Glial swelling in ischemia: A hypothesis. Dev. Neurosci. 15:216–225
Author information
Authors and Affiliations
Additional information
I wish to express my gratitude to Mme J. Verner for her expert assistance during this project and to my colleague Dr. R. Sauvé for reading and commenting on the manuscript. Financial support was provided by the Natural Sciences and Engineering Council of Canada and by the “Fonds pour la Formation de Chercheur et l'Activité de la Recherche” of Quebec.
Rights and permissions
About this article
Cite this article
Roy, G. Amino acid current through anion channels in cultured human glial cells. J. Membarin Biol. 147, 35–44 (1995). https://doi.org/10.1007/BF00235396
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00235396