Summary
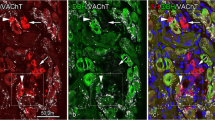
The distribution of serotonin immunoreactive (5-HT-IR) fibers in the trigeminal nuclear complex of the rat was mapped. In the sensory nuclei, innervation appeared to be dense in areas primarily related to nociceptive afferent activity, and sparse in areas primarily related to nonnociceptive afferent activity. Specifically, the marginal and gelatinosa layers of the spinal subnucleus caudalis contained many 5-HT-IR fibers while few labeled fibers were seen in the magnocellular portion of subnucleus caudalis or in the principal sensory nucleus. The spinal subnuclei oralis and interpolaris were sparsely innervated except for a few areas which contained more 5-HT-IR fibers. The motor nucleus contained as many immunoreactive fibers as the subnucleus caudalis, although fibers in the motor nucleus were thicker and varicosities more irregularly spaced than in caudalis.
Similar content being viewed by others
References
Azerad J, Woda A, Albe-Fessard D (1982) Physiological properties of neurons in different parts of the cat trigeminal sensory complex. Brain Res 246: 7–21
Burns LL, Haigler HJ (1983) Effects of serotonin and morphine on spontaneous and evoked firing of nociceptive neurons in the trigeminal spinal nucleus of rats. Exp Neurol 79: 688–703
Chan-Palay V (1978) Indoleamine neurons and their processes in the normal rat brain and in chronic diet-induced thiamine deficiency demonstrated by uptake of 3H-serotonin. J Comp Neurol 176: 467–494
Cropper EC, Eisenman JS, Azmitia E (1983) An immunocytochemical study of the serotonergic innervation of the trigeminal nuclear complex in the rat. Proc Soc Neurosci 9: 727 (Abstract)
DeSantis M, Limwongse V, Rigamonti D (1978) Somatotopy in the trigeminal motor nucleus of the rat: Field potentials recorded in the neuron pool after retrograde transport of horseradish peroxidase. Neurosci Lett 10: 95–98
Eisenman J, Landgren S, Novin D (1963) Functional organization in the main sensory trigeminal nucleus and in the rostral subdivision of the nucleus of the spinal trigeminal tract in the cat. Acta Physiol Scand 59 Suppl 214: 1–44
Erzurumlu RS, Bates CA, Killackey HP (1980) Differential organization of thalamic projection cells in the brain stem trigeminal complex of the rat. Brain Res 198: 427–433
Frankfurt M, Lauder JM, Azmitia EC (1981) The immunocytochemical localization of serotonergic neurons in the rat hypothalamus. Neurosci Lett 24: 227–232
Fukushima T, Kerr FWL (1979) Organization of trigeminothalamic tracts and other thalamic afferent systems in the brainstem of the rat: Presence of gelatinosa neurons with thalamic connections. J Comp Neurol 183: 169–184
Fuxe K (1965) Evidence for the existence of monoamine neurons in the central nervous system. IV. The distribution of monoamine terminals in the central nervous system. Acta Physiol Scand 64 Suppl 247: 39–85
Kirkpatrick DB, Kruger L (1975) Physiological properties of neurons in the principal sensory trigeminal nucleus of the cat. Exp Neurol 48: 664–690
Lauder JM, Petrusz P, Wallace JA, Dinome A, Wilkie M, McCarthy K (1982) Combined serotonin immunocytochemistry and 3H-thymidine autoradiography: In vivo and in vitro methods. J Histochem Cytochem 30: 788–793
Limwongse V, DeSantis M (1977) Cell body locations and axonal pathways of neurons innervating muscles of mastication in the rat. Am J Anat 149: 477–488
McCall RB, Aghajanian GK (1973) Serotonergic facilitation of facial motoneuron excitation. Brain Res 169: 11–27
Mosso JA, Kruger L (1973) Receptor categories represented in spinal trigeminal nucleus caudalis. J Neurophysiol 36: 472–488
Olszewski J (1950) On the anatomical and functional organization of the spinal trigeminal nucleus. J Comp Neurol 92: 401–413
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic Press, Sydney
Price DD, Dubner R, Hu JW (1976) Trigeminothalamic neurons in nucleus caudalis responsive to tactile, thermal, and nociceptive stimulation of monkey's face. J Neurophysiol 39: 936–953
Rogawski MA, Aghajanian GK (1980) Norepinephrine and serotonin: Opposite effects on the activity of lateral geniculate neurons evoked by optic pathway stimulation. Exp Neurol 69: 678–694
Ruda MA, Gobel S (1980) Ultrastructural characterization of axonal endings in the substantia gelatinosa which take up [3H]serotonin. Brain Res 184: 57–83
Shigenaga Y, Takabatake M, Sugimoto T, Sakai A (1979) Neurons in marginal layer of trigeminal nucleus caudalis projecting to ventrobasal complex (VB) and posterior nuclear complex (PO) demonstrated by retrograde labeling with horseradish peroxidase. Brain Res 166: 391–396
Smith RL (1973) The ascending fiber projections from the principal sensory trigeminal nucleus in the rat. J Comp Neurol 148: 423–446
Steinbusch HWM (1981) Distribution of serotonin-immunoreactivity in the central nervous system of the rat — cell bodiesand terminals. Neuroscience 6: 557–618
Steinbusch HWM, Verhofstad AAJ, Joosten HWJ (1978) Localization of serotonin in the central nervous system by immunocytochemistry: Description of a specific and sensitive technique and some applications. Neuroscience 3: 811–819
Sternberger LA (1979) Immunocytochemistry (2nd ed). John Wiley and Sons, New York, pp 104–169
Wallace JA, Petrusz P, Lauder JM (1982) Serotonin immunocytochemistry in the adult and developing rat brain: Methodological and pharmacological considerations. Brain Res Bull 9: 117–129
Willis WD (1981) Effects of peripherally and centrally administered serotonin on primate spinothalamic neurons. In: Haber B et al. (eds) Serotonin: Current aspects of neurochemistry and function. Plenum Press, New York (Advances in experimental medicine and biology, vol 133, pp 105–123)
VanderMaelen CP, Aghajanian GK (1980) Intracellular studies showing modulation of facial motoneurone excitability by serotonin. Nature 287: 346–347
Author information
Authors and Affiliations
Additional information
This research was supported by grant BNS-79-06474 from the National Science Foundation awarded to Efrain Azmitia
Rights and permissions
About this article
Cite this article
Cropper, E.C., Eisenman, J.S. & Azmitia, E.C. 5-HT-immunoreactive fibers in the trigeminal nuclear complex of the rat. Exp Brain Res 55, 515–522 (1984). https://doi.org/10.1007/BF00235282
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00235282