Abstract
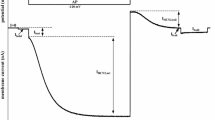
Using the two-microelectrode voltage clamp technique in Xenopus laevis oocytes, we estimated Na+-K+-ATPase activity from the dihydroouabain-sensitive current (I DHO) in the presence of increasing concentrations of tetraethylammonium (TEA+; 0, 5, 10, 20, 40 mm), a well-known blocker of K+ channels. The effects of TEA+ on the total oocyte currents could be separated into two distinct parts: generation of a nonsaturating inward current increasing with negative membrane potentials (V M) and a saturable inhibitory component affecting an outward current easily detectable at positive V M. The nonsaturating component appears to be a barium-sensitive electrodiffusion of TEA+ which can be described by the Goldman-Hodgkin-Katz equation, while the saturating component is consistent with the expected blocking effect of TEA+ on K+ channels. Interestingly, this latter component disappears when the Na+-K+-ATPase is inhibited by 10 μm DHO. Conversely, TEA+ inhibits a component of I DHO with a k d of 25±4 mm at +50 mV. As the TEA+-sensitive current present in I DHO reversed at −75 mV, we hypothesized that it could come from an inhibition of K+ channels whose activity varies in parallel with the Na+-K+-ATPase activity. Supporting this hypothesis, the inward portion of this TEA+-sensitive current can be completely abolished by the addition of 1 mm Ba2+ to the bath. This study suggests that, in X. laevis oocytes, a close link exists between the Na-K-ATPase activity and TEA+-sensitive K+ currents and indicates that, in the absence of effective K+ channel inhibitors, I DHO does not exclusively represent the Na+-K+-ATPase-generated current.
Similar content being viewed by others
References
Apell, H.J. 1989. Electrogenic properties of the Na,K pump. J. Membrane Biol. 110:103–114
Bahinski, A., Nakao, M., Gadsby, D.C. 1988. Potassium translocation by the Na+/K+ pump is voltage insensitive. Proc. Natl. Acad. Sci. USA 85:3412–3416
Beck, J.S., Hurst, A.M., Lapointe, J.-Y., Laprade, R. 1993. Regulation of basolateral K channels in proximal tubule studied during continuous microperfusion. Am. J. Physiol. 264:F496-F501
Beck, J.S., Laprade, R., Lapointe, J.-Y. 1994. Coupling between transepithelial Na transport and basolateral K conductance in the renal proximal tubule. Am. J. Physiol. 266:F517-F527
Dascal, N. 1987. The use of Xenopus oocytes for the study of ion channels. CRC, Crit. Rev. Biochem. 22:317–386
De Weer, P. 1992. Cellular sodium-potassium transport. In: The Kidney:Physiology and Pathophysiology. D.W. Seldin and G. Giebisch, editors. Second edition, pp. 93–112. Raven, New York
Dumont, J.N. 1972. Oogenesis in Xenopus laevis (Daudin) I. Stages of oocyte development in laboratory maintained animals. J. Morphol. 136:153–180
Gadsby, D.C., Rakowski, R.F., De Weer, P. 1993. Extracellular access to the Na, K pump: pathway similar to ion channel. Science 260:100–103
Hermann, A., Gorman, A.L.F. 1981. Effects of tetraethylammonium on potassium currents in a molluscan neuron. J. Gen. Physiol. 78:87–110
Hilgeman, D.W. 1994. Channel-like function of the Na,K pump probed at microsecond resolution in giant patches. Science 263:1429–1432
Hille, B. 1970. Ionic channels in nerve membranes. Prog. Biophys. Mol. Biol. 21:3–32
Hurst, A.M., Beck, J.S., Laprade, R., Lapointe, J.-Y. 1993. Na+ pump inhibition downregulates an ATP-sensitive K+ channel in rabbit proximal convoluted tubule. Am. J. Physiol. 264:F760-F764
Lafaire, A.V., Schwarz, W. 1986. Voltage dependence of the rheogenic Na+/K+ ATPase in the membrane of oocytes of Xenopus laevis. J. Membrane Biol. 91:43–51
Lang, F., Messner, G., Rehwald, W. 1986. Electrophysiology of sodium-coupled transport in proximal renal tubules. Am. J. Physiol. 250:F953-F962
Nakao, M., Gadsby, D.C. 1986. Voltage dependence of Na translocation by the Na/K pump. Nature 323:628–630
Neher, E., Lux, H.D. 1972. Differential action of TEA+ on two K+-current components of a molluscan neurone. Pfluegers Arch. 336:87–100
Peres, A., Bernardini, G., Mancinelli, E., Ferroni, A. 1985. A voltage-dependent K+ channel controlling the membrane potential in frog oocytes. Pfluegers Arch. 403:41–46
Rakowski, R.F., Paxson, C.L. 1988. Voltage dependence of Na/K pump current in Xenopus oocytes. J. Membrane Biol. 106:173–182
Rakowski, R.F., Vasilet, L.A., La Tona, J., Schwarz, W. 1991. A negative slope in the current-voltage relationship of the Na+/K+ pump in Xenopus oocytes produced by reduction of external [K+]. J. Membrane Biol. 121:177–187
Rakowski, R.F. 1993. Charge movement by the Na/K pump in Xenopus oocytes. J. Gen. Physiol. 101:117–144
Sachs, J.R., Conrad, M.E. 1968. Effect of tetraethylammonium on the active cation transport system of the red blood cell. Am. J. Physiol. 215:795–798
Schultz, S.G. 1981. Homocellular regulatory mechanisms in sodium-transporting epithelia: avoidance of extinction by “flushthrough”. Am. J. Physiol. 241:F579-F590
Schweigert, B., Lafaire, A.V., Schwarz, W. 1988. Voltage dependence of the Na-K ATPase: measurements of ouabain in-dependent membrane current and ouabain binding in oocytes of Xenopus laevis. Pfluegers Arch. 412:579–588
Sturmer, W., Buhler, R., Apell, H.-J., Laüger, P. 1991. Charge translocation by the Na,K pump: II. Ion binding and release at the extracellular face. J. Membrane Biol. 121:163–176
Thompson, S.H. 1977. Three pharmacologically distinct potassium channels in molluscan neurones. J. Physiol. 265:465–488
Vasilet, L.A., Schwarz, W. 1992. Regulation of endogenous and expressed Na+/K+ pumps in Xenopus oocytes by membrane potential and stimulation of protein kinases. J. Membrane Biol. 125:119–132
Wu, M.M., Civan, M.M. 1991. Voltage dependence of current through the Na,K-exchange pump of Rana oocytes. J. Membrane Biol. 121:23–36
Zemkova, H., Teisinger, J., Vyskocil, F. 1988. Inhibition of the electrogenic Na,K pump and Na,K-ATPase activity by tetraethylammonium, tetrabutylammonium, and apamin. J. Neurosci. Res. 19:497–503
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Huang, H., St.-Jean, H., Coady, M.J. et al. Evidence for coupling between Na+ pump activity and TEA-sensitive K+ currents in Xenopus laevis oocytes. J. Membarin Biol. 143, 29–35 (1995). https://doi.org/10.1007/BF00232521
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232521