Summary
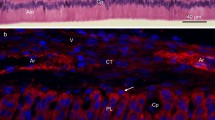
Repeated injection of rats with tunicamycin over two days induced a 1- to 5-fold increase in glycogen. This accumulation occurred in the stratum intermedium of the enamel organ and in young secretory odontoblasts. In rats injected over 3 days, the number of glycogen particles was at least 10 times larger than in control rats, and large glycogen accumulations were observed in the cytosol of these two groups of cells. These results were obtained by staining with periodic acid-thiocarbohydrazide and silver proteinate, a specific method for the detection of glycoconjugates containing vic-glycol groups. The existence of a relationship between these local cytosolic accumulations of glycogen and the developmental stage of certain groups of cells was shown by the changes that occurred in glycogen distribution. The present results suggest that the stratum intermedium supplies energy for precursor transport.
Similar content being viewed by others
References
Butler WT (1984) Matrix macromolecules of bone and dentine. Coll Relat Res 4:297–307
Duksin D, Bornstein P (1977) Changes in surface properties of normal and transformed cells caused by tunicamycin, an inhibitor of protein glycosylation. Proc Natl Acad Sci USA 74:3433–3437
Goldberg M, Escaig F (1982) Epithelium adamantin externe, réticulum étoile et stratum intermedium de l'organe de l'émail de l'incisive chez le rat: étude ultrastructurale sur coupe et aprés cryofracture. J Biol Buccale 10:293–306
Goldberg M, Escaig F, Septier D (1982) The effects of vinblastine on the cell structure and activity in the rat incisor enamel organ during the secretory stage “in vivo” as shown by autoradiography using 3H-proline, 3H-serine and 3H-fucose. J Biol Buccale 10:237–259
Heifetz A, Keenan RW, Elbein AD (1979) Mechanism of action of tunicamycin on the UDP-GlcNAc: dolichyl-phosphate GlcNAc-1-phosphate transferase. Biochemistry 18:2186–2192
Hickman S, Kulczycki A, Lynch RG, Kornfeld S (1977) Studies of the mechanism of tunicamycin inhibition of IgA and IgE secretion by plasma cells. J Biol Chem 252:4402–4408
Ishimura K, Kurihara H, Fujita H (1984) Effects of tunicamycin on thin-section and freeze-fracture images of microvilli of the duodenal epithelial cells of the mouse. Cell Tissue Res 238:653–565
Kallenbach E (1978) Fine structure of the stratum intermedium, stellate reticulum and outer enamel epithelium in the enamel organ of the kitten. J Anat 126:247–260
Kitagawa K, Nishino H, Iwashima A (1985) Effect of tunicamycin on hexose transport in mouse embryo fibroblast Swiss 3T3 cells. Biochim Biophys Acta 821:67–71
Lehle L, Tanner W (1976) The specific site of tunicamycin inhibition in the formation of dolichol-bound N-acetylglucosamine derivatives. FEBS Lett 71:167–170
Linde A (1984) Non-collagenous proteins and proteoglycans in dentinogenesis. In: Linde A (ed) Dentin and Dentinogenesis, Vol II., CRC Press, Inc Boca Raton, Florida, pp 55–92
Michaels JE (1980) Alterations in the distribution of glycoproteins in epithelial cells of murine colon after injection of tunicamycin. Cell Tissue Res 210:121–132
Nishikawa Y, Yamamoto Y, Onodera K, Tamura G, Mitoui H (1979) Altered molecular structure of HLA-DR antigens synthesized in the presence of tunicamycin. Biochem Biophys Res Commun 87:1235–1242
Olden K, Pratt RM, Yamada KM (1978) Role of carbohydrates in protein secretion and turnover: effects of tunicamycin on the major cell surface glycoproteins of chick embryo fibroblasts. Cell 13:461–473
Orams HJ, Snibson KJ (1982) Ultrastructural localization and gradient of activity of alkaline phosphatase activity during rodent odontogenesis. Calcif Tissue Int 34:273–279
Ream LJ, Principato R (1981) Glycogen accumulation in the parathyroid gland of the rat after fluoride ingestion. Cell Tissue Res 220:125–130
Reith EJ, Butcher EO (1967) Microanatomy and histochemistry of amelogenesis. In: Miles AEW (ed) Structural and chemical organization of teeth, Vol I., Academic Press, New York London, pp 371–397
Symons NBB (1967) The microanatomy and histochemistry of dentinogenesis. In: Miles AEW (ed) Structural and chemical organization of teeth, Vol I, Academic Press, New York London, pp 285–324
Takatsuki A, Arima K, Tamura G (1971) Tunicamycin, a new antibiotic. I. Isolation and characterization of tunicamycin. J Antibiot 24:215–223
Termine JD, Belcourt AB, Christner PJ, Conn KM, Nylen MU (1980) Properties of dissociatively extracted fetal tooth matrix proteins. I. Principal molecular species in developing bovine enamel. J Biol Chem 225:9760–9768
Thiery JP (1967) Mise en évidence des polysaccharides sur coupes fines en microscopie électronique. J Microscopie 6:987–1018
Thiery JP (1969) Róle de l'appareil de Golgi dans la synthése des mucopolysaccharides. Etude cytochimique. I. Mise en évidence de mucopolysaccharides dans les vésicules de transition entre l'ergastoglasme et l'appareil de Golgi. J Microscopie 8:689–703
Waechter CJ, Harford JB (1977) Evidence of the enzymatic transfer of N-acetylglycosamine from UDP-N-acetyl glucosamine into dolichol derivatives and glycoproteins by calf brain membranes. Arch Biochem Biophys 181:185–198
Waechter CJ, Lennarz WJ (1976) The role of polyprenol-linked sugars in glycoprotein synthesis. Annu Rev Biochem 45:95–112
Wislocki GB, Singer M, Waldo CM (1948) Some histochemical reaction of mucopolysaccharides, glycogen, lipids and other substances in teeth. Anat Rec 101:487–514
Yanagishita M (1986) Tunicamycin inhibits proteoglycan synthesis in rat ovarian granulosa cells in culture. Arch Biochem Biophys 251:287–298
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chardin, H., Septier, D., Lecolle, S. et al. Effect of tunicamycin on glycogen accumulation in the stratum intermedium and odontoblasts of rat incisor. Cell Tissue Res. 256, 519–527 (1989). https://doi.org/10.1007/BF00225600
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00225600