Summary
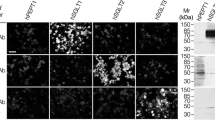
An antibody to the 96 kD α-subunit of the Na+, K+ -ATPase from Bufo marinus has been used in immunostaining rat kidney and salivary glands. Intense staining was observed on basolateral membranes of distal tubules of the kidney and striated ducts of the three major salivary glands. Less intense staining was seen on the basolateral membranes of parotid acinar cells, but no staining was seen on the acinar cells of submandibular or sublingual glands. These sites of staining have been shown, by other methods, to posses substantial Na+, K+ -ATPase, indicating that the antibody recognizes antigenic determinants of the sodium pump highly conserved in the course of evolution. In addition, staining with this antibody was observed at the apical region of cells of the proximal straight tubule and of the papillary collecting duct in the kidney. Absorption studies suggest that the apical antigenic determinants are the same or closely related to each other but are distinct from basolateral antigenic determinants.
Similar content being viewed by others
References
Baskin DG, Stahl WL (1982) Immunocytochemical localization of Na+ K+-ATPase in the rat kidney. Histochemistry 73:535–548
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Ernst SA, Hootman SR (1981) Microscopical methods for the localization of Na+, K+ -ATPase. Histochem J 13:397–418
Ernst SA, Palacios JR II, Siegel GJ (1986) Immunocytochemical localization of Na+, K+ -ATPase catalytic polypeptide in mouse choroid plexus. J Histochem Cytochem 34:189–195
Fambrough DM, Bayne EK (1983) Multiple forms of (Na++K+)-ATPase in the chicken. Selective detection of the major nerve, skeletal muscle and kidney form by a monoclonal antibody. J Biol Chem 258:3926–3935
Geering K, Girardet M, Bron C, Kraehenbühl J-P, Rossier BC (1982) Hormonal regulation of (N+, K+)-ATPase biosynthesis in the toad bladder. Effect of aldosterone and 3,5,3-triiodo-L-thyronine. J Biol Chem 257:10338–10343
Geering K, Meyer DI, Paccolat M-P, Kraehenbühl J-P, Rossier BC (1985) Membrane insertion of α-and β-subunits of Na+, K+ -ATPase. J Biol Chem 260:5154–5156
Geering K, Kraehenbuhl J-P, Rossier BC (1987) Maturation of the catalytic α-subunit of Na, K-ATPase during intracellular transport. J Cell Biol 105:2613–2619
Girardet M, Geering K, Frantes JM, Geser D, Rossier BC, Kraehenbühl J-P, Bron C (1981) Immunochemical evidence for a transmembrane orientation of both the (Na+, K+)-ATPase subunits. Biochemistry 20:6684–6691
Hootman SR (1986) Neuroendocrine control of secretion in pancreatic and parotid gland acini and the role of Na+, K+-AT-Pase activity. Int Rev Cytol 105:129–181
Jørgensen PL (1980) Sodium and potassium ion pump in kidney tubules. Physiol Rev 60:864–917
Jørgensen PL (1986) Structure, function and regulation of Na, K-ATPase in the kidney. Kidney Int 39:10–20
Katz AI (1982) Renal Na-K-ATPase: its role in tubular sodium and potassium transport. Am J Physiol 242:F207-F219
Kriz W, Bankir L (1988) A standard nomenclature for structures of the kidney. Am J Physiol 254:F1-F8
Shull GE, Schwartz A, Lingrel JB (1985) Amino-acid sequence of the catalytic subunit of the (Na+ K+) ATPase deduced from a complementary DNA. Nature 316:691–695
Siegel GJ, Holm C, Schreiber JH, Desmond T, Ernst SA (1984) Purification of mouse brain (Na++K+)-ATPase catalytic unit, characterization of antiserum, and immunocytochemical localization in cerebellum, choroid plexus and kidney. J Histochem Cytochem 32:1309–1318
Siegel GJ, Desmond T, Ernst SA (1986) Immunoreactivity and ouabain-dependent phosphorylation of (Na++K+)-adenosinetriphosphatase catalytic subunit doublets. J Biol Chem 261:13768–13776
Simson JAV, Spicer SS, Chao J, Grimm L, Margolius HS (1979) Kallikrein localization in rodent salivary glands and kidney with the immunoglobulin-enzyme bridge technique. J Histochem Cytochem 27:1567–1576
Speight PM, Chisholm DM (1984) Tee relationship between localization of Na+, K+-ATPase and cellular fine structure in the rat parotid gland. Histochem J 16:721–731
Tamarin A, Sreebny LM (1965) The rat submaxillary salivary gland. A correlative study by light and electron microscopy. J Morphol 117:295–352
Winston DC, Hennigar RA, Spicer SS, Garrett JR, Schulte BA (1988) Immunohistochemical localization of NA+, K+-ATPase in rodent and human salivary and lacrimal glands. J Histochem Cytochem 36:1139–1145
Young JA, van Lennep EW (1979) Transport in salivary and salt glands. In: Giebisch G, Tosteson DC, Ussing HH (eds) Membrane Transport in Biology, Vol. IV B. Springer, Berlin Heidelberg New York, pp 563–574
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Graves, J.S., Inabnett, T., Geering, K. et al. Cross-reactivity of an antiserum to the α-subunit of the Na+, K+-ATPase of toad (Bufo marinus) kidney with basal and apical membranes of transporting epithelia of the rat. Cell Tissue Res. 258, 137–145 (1989). https://doi.org/10.1007/BF00223153
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00223153