Summary
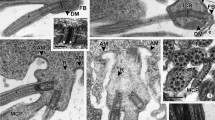
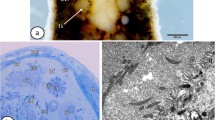
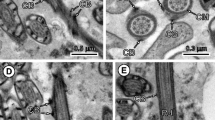
Typosyllis pulchra reproduces by the production of three to four gamete-bearing stolons (schizogamy) during consecutive 30-day periods. Although gonads are found in a large number of segments, only those in the posterior-most segments produce gametes and become incorporated into the developing stolon. The more anterior gonads remain undifferentiated and probably sexually undetermined until they are needed in future stolonizations. Gonial cells, which will eventually become either male or female, are ultrastructurally identical at the onset of each stolonization period. Spermatogenesis is marked by a short proliferative period followed by differentiation and spermiogenesis. The first ultrastructural signs of spermatogenesis were found in coelomic spermatogonia on day 10 of stolon formation. Spermatogonia are joined by intercellular bridges, which are maintained until the early spermatid stage. Synaptonemal complexes mark the onset of meiosis, which is apparently synchronized in the syncytial clusters of primary spermatocytes. Spermiogenesis occurs during the final 10 days of stolonization and a variety of stages is present within a single animal. All sperm mature by the time the stolon detaches. Acrosome formation and nuclear condensation are described in addition to the ultrastructure of mature sperm.
Similar content being viewed by others
Reference
Anderson TF (1951) Techniques for the preservation of three-dimensional structure in preparing specimens for the electron microscope. Trans NY Acad Sci 13:130–133
Bertout M (1976) Spermatogenèse de Nereis diversicolor OF Muller (Annélide Polychète). 1 Évolution du cytoplasme et élaboration de l'acrosome. J Microscopie Biol Cell 25:87–94
Bertout M (1977) Spermatogenèse de Nereis diversicolor (Muller OF) (Annélide Polychète). 2 Étude morphologique et autoradiographique de l'évolution du noyau. Biol Cell 30:211–216
Cloney RA, Florey E (1968) Ultrastructure of cephalopod chromatophore organs. Z Zellforsch 89:250–280
Colwin AL, Colwin LH (1961a) Fine structure of the spermatozoon of Hydroides hexogonus (Annelida) with special reference to the acrosomal region. J Biophys Biochem Cytol 10:211–230
Colwin LH, Colwin AL (1961b) Changes in the spermatozoon during fertilization in Hydroides hexagonus (Annelida). I Passage of the acrosomal region through the vitelline membrane. J Biophys Biochem Cytol 10:231–254
Colwin LH, Colwing AL (1961c) Changes in the spermatozoon during fertilization in Hydroides hexagonus (Annelida). II Incorporation with the egg. J Biophys Biochem Cytol 10:255–274
Cotelli F, Lora Lamia Dinin L (1975) Ultrastructural analysis of mature spermatozoa of Hyalinoecia tubicola (Muller OF) (Annelidae Polychaeta). Monitore Zool Ital 9:51–66
Daly JM (1975) Reversible epitoky in the life history of the polychaete Odontosyllis polycera (Schmarda 1861). J Mar Biol Ass UK 55:327–344
Durchon M (1951) Stolonisation et hemaphroditisme successif chez Syllis amica Quatrefages. Arch Zool Exp Gen 88:96–100
Durchon M (1959) Contribution à l'étude de la Stolonisation chez les Syllidiens (Annélides Polychètes). I. Syllinae. Bull Biol Fr Belg 93:155–219
Eckelbarger KJ (1975) A light and electron microscope investigation of gametogenesis in Nicolea zostericola (Grube 1860) (Polychaeta: Terebellidae). Mar Biol 31:116–134
Eckelbarger KJ (1979) Ultrastructural evidence for both autosynthetic and heterosynthetic yolk formation in the oocytes of an annelid (Phragmatopoma lapidosa: Polychaeta). Tissue and Cell 11:425–443
Fallin JF, Austin CR (1967) Fine structure of gametes of Nereis limbata (Annelida) before and after interaction. J Exp Zool 166:225–242
Fawcett DW (1961) Intercellular bridges. Exp Cell Res 22: Suppl 8:174–187
Franke HD Experimentelle Untersuchungen zur Steuerung des Fortpflanzungrhythmus und der sexuellen Differenzierung an Laborkulturen des Polychaeten Typosyllis prolifera (Krohn 1852). Dissertation, Technische Universität zu Braunschweig
Franzen A (1956) On spermiogenesis, morphology of the spermatozoon, and biology of fertilization among invertebrates. Zool Bidr Uppsala 31:355–482
Franzen A (1974) Sperm ultrastructure in some polychaeta, p 267–278. In: Afzelius BA (ed) The functional anatomy of the spermatozoon. Pergamon Press, New York
Franzen A (1975) Fine structure of spermiogenesis in Fabricia sabella (Ehrenberg), Polychaeta Family Sabellidae Zoon 3, 1–10
Gidholm L (1963) The sexual organs in the budding form of Autolytus. Zool Bidr Uppsala 35:529–544
Graebner I, Kryvi H (1973) The spermiogenesis and mature sperm of Sabella penicillum (Polychaeta). An electron microscopical investigation. Norw J Zool 21:211–226
Heacox AE (1980) Reproduction, ultrastructure of gametogenesis and endocrine control of gametogenesis and sex determination in Typosyllis pulchra (Polychaeta: Syllidae) Dissertation, Washington State University, Pullman
Humphrey DC, Pittman FE (1974) A simple methylene blue-azure II-basic fuchsin stain for epoxyembedded tissue sections. Stain Technol 49:9–14
Kubo M, Sawada N (1977) Electron microscope study on sperm differentiation in Perinereis brevicirris (Polychaeta). Cell Struct Funct 2:135–144
Kubo M, Ishikawa M (1978) Organizing process of the temporary acrosome in spermatogenesis of the bivalve Lyonsia ventricola. J Submicr Cytol 10:411–421
Longo JF, Anderson E (1969) Spermiogenesis in the surf clam, Spisula solidissima with special reference to the formation of the acrosomal vesicle. J Ultrastruct Res 27:435–443 (1969)
Longo JF, Anderson E (1974) Gametogenesis. In: Lash J, Whittaker JR (eds) Concepts of development. Sinauer Associates Stanford, p 3–47
Longo JF, Dornfield EJ (1967) The fine structure of spermatid differentiation in the mussel, Mytilus edulis. J Ultrastruct Res 20:426–480
Luft JH (1961) Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol 9:409–414
Malaquin A (1893) Recherches sur les Syllidiens. Morphologie, anatomie, réproduction, developpement. Mem Soc Sci Arts Lille, pp 1–477
Olive PJW, Clark RB (1978) Physiology of reproduction. In: Mill PJ (ed) Physiology of Annelids. Academic Press, New York
Potswald HE (1967) An electron microscope study of spermiogenesis in Spirorbis (Laeospira) morchi. Biol Bull 135:208–222
Potswald HE (1972) The relationship of early oocytes to putative neoblasts in the serpulid Spirorbis borealis. J Morphol 135:215–229
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Roosen-Runge EC (1977) The process of spermatogenesis in animals. Cambridge University Press, Cambridge, p 1–214
Sawada N (1975) Electron microscope studies of sperm differentiation in marine annelid worms. II. Sperm formation in Arenicola brasiliensis. Devel Growth Differ 17:89–99
Schroeder PC, Hermans CO (1975) Annelida: Polychaeta In: Geise AC, Pearse JS (eds) Reproduction of Marine Invertebrates, Vol 3, 1–213, Annelids and Echiurans. Academic Press, New York
Wissocq JC (1966) La sexualisation du stolon chez Syllis spongicola Grube. Cah Biol Mar 7:337–342
Wynne-Edwards VC (1962) Animal dispersion in relation to social behavior. Oliver And Boyd, London, p 1–653
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heacox, A.E., Schroeder, P.C. A light- and electron-microscopic investigation of gametogenesis in Typosyllis pulchra (Berkeley and Berkeley) (polychaeta: Syllidae). Cell Tissue Res. 218, 623–639 (1981). https://doi.org/10.1007/BF00210120
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00210120