Abstract
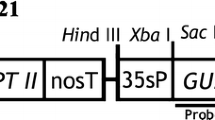
Transgenic tobacco (Nicotiana tabacum L.), licorice (Glycyrrhiza uralensis Fisher) and foxglove (Digitalis purpurea L.) were obtained with binary vector systems based on a disarmed Agrobacterium tumefaciens and on a virulent A. rhizogenes. The chimeric neomycin phosphotransferase II (NPT-II) gene (kan) and the ß-glucuronidase (GUS) gene (uidA) were under the control of the TR1′ and 2′ promoters, respectively, on a binary vector, pGSGluc1. Tissue-specific expression of the chimeric TR2' -uidA gene was studied using regenerants of N. tabacum from transformed callus and hairy roots, and also using the transformed roots of G. uralensis and D. purpurea. In all these transformed tissues, the phloem and surrounding tissues were stained with 5-bromo-4-chloro-3-indolyl-ß-d-glucuronide, showing the specific expression of uidA. The enzymatic assays of NPT-II and GUS indicated that the expression of both TR1' -kan and TR2'- uidA were coordinately enhanced by wounding and by the addition of plant growth regulators. These results indicate that the gene expression by the dual TR promoters is regulated in the several plant species studied in a tissue-specific manner and enhanced by physiological stresses.
Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- GUS:
-
β-glucuronidase
- Km = kanamycin:
-
NPT-II neomycin phosphotransferase II
- Rif:
-
rifampicin
- X-gluc:
-
5-bromo-4-chloro-3-indolyl-β-d-glucuronide
References
Benfey, P.N., Chua, N.-H. (1989) Regulated genes in transgenic plants. Science 244, 174–181
Bevan, M., Schufflebottom, D., Edwards, K., Jefferson, R., Schuch, W. (1989) Tissue- and cell-specific activity of a phenylalanine ammonia-lyase promoter in transgenic plants. EMBO J. 8, 1899–1906
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254
Deblaere, R., Bytebier, B., De Greve, H., Deboeck, F., Schell, J., Van Montagu, M., Leemans, J. (1985) Efficient octopine Ti plasmid-derived vectors for Agrobacterium-mediated gene transfer to plants. Nucleic Acids Res. 13, 4777–4788
Deblaere, R., Reynaerts, A., Höfte, H., Hernalsteens, J.-P., Leemans, J., Van Montagu, M. (1987) Vectors for cloning in plant cells. Methods in Enzymol. 153, 277–292
De Block, M., Botterman, J., Vandewiele, M., Dockx, J., Thoen, C., Gossele, V., Rao Movva, N., Thompson, C., Van Montagu, M., Leemans, J. (1987) Engineering herbicide resistance in plants by expression of a detoxifying enzyme. EMBO J. 6, 2513–2518
Dekeyser R., Claes B., Marichal, M., Van Montagu, M., Caplan, A. (1989) Evaluation of selectable markers for rice transformation. Plant Physiol. 90, 217–223
Dellaporta, S.L., Wood, J., Hicks, J.B. (1983) A plant DNA minipreparation: version II. Plant Mol. Biol. Rep. 1, 19–21
Figurski, D.H., Helinski, D.R. (1979) Replication of an origincontaining derivative of pRK2 dependent on a plasmid function provided in trans. Proc. Natl. Acad. Sci. USA 76, 1648–1652
Gamborg, O.L., Miller, R.A., Ojima, K. (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell Res. 50, 151–158
Gasser, C.S., Fraley, R.T. (1989) Genetically engineering plants for crop improvement. Science 244, 1293–1299
Harpster M.H., Townsend, J.A., Jones, J.D.G., Bedbrook, J., Dunsmuir, P. (1988) Relative strengths of the 35S cauliflower mosaic virus, 1', 2', and nopaline synthase promoters in transformed tobacco, sugarbeet and oilseed rape callus tissue. Mol. Gen. Genet. 212, 182–190
Jefferson, R.A., Kavanagh, T.A., Bevan, M. (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6, 3901–3907
Jouanin, L. (1984) Restriction map of an agropine-type Ri plasmid and its homologies with Ti plasmids. Plasmid 12, 91–102
Langridge, W.H.R., Fitzgerald, K.J., Koncz, C., Schell, J., Szalay, A.A. (1989) Dual promoter of Agrobacterium tumefaciens mannopine synthase genes is regulated by plant growth hormones. Proc. Natl. Acad. Sci. USA 86, 3219–3223
Murashige, T., Skoog, F. (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 15, 473–479
Neumann, D. (1985) Storage of alkaloids. In: Biochemistry of alkaloids, pp. 49–55, Mothes, K., Schütte, H.R., Luckner, M., eds. VCH Publishers, Weinheim, FRG
Peleman, J., Wout, B., Engler, G., Seurinck, J., Botterman, J., Alliotte, T., Van Montagu, M., Inzé, D. (1989a) Strong cellular preference in the expression of a housekeeping gene of Arabidopsis thaliana encoding S-adenosylmethionine synthetase. Plant Cell 1, 81–93
Peleman, J., Saito, K., Cottyn, B., Engler, G., Seurinck, J., Van Montagu, M., Inzé, D. (1989b) Structure and expression analysis of the S-adenosylmethionine synthetase gene family in Arabidopsis thaliana. Gene 84, 359–369
Roger, S.G., Klee, H. (1987) Pathways to plant genetic manipulation employing Agrobacterium. In: Plant DNA infectious agents, pp. 178–203, Hohn, T., Schell, J., eds. Springer, Wien, New York
Saito, K., Murakoshi, I., Inzé, D., Van Montagu, M. (1989a) Biotransformation nicotine alkaloids by tobacco shooty teratomas induced by a Ti plasmid mutant. Plant Cell Rep. 7, 607–610
Saito, K., Yamazaki, M., Takamatsu, S., Kawaguchi, A., Murakoshi, I. (1989b) Greening induced production of (+)-lupanine in tissue culture of Thermopsis lupinoides. Phytochemistry 28, 2341–2344
Saito, K., Yamazaki, M., Yamakawa, K., Fujisawa, S., Takamatsu, S., Kawaguchi, S., Murakoshi, I. (1989c) Lupin alkaloids in tissue culture of Sophora flavescens var. angustifolia: Greening induced production of matrine. Chem. Pharm. Bull. 37, 3001–3004
Saito, K., Kaneko, H., Yamazaki, M., Yoshida, M., Murakoshi, I. (1990a) Stable transfer and expression of chimeric genes in licorice (Glycyrrhiza uralensis) using an Ri plasmid binary vector. Plant Cell Rep. 8, 718–721
Saito, K., Yamazaki, M., Shimomura, K., Yoshimatsu, K., Murakoshi, I. (1990b) Genetic transformation of foxglove (Digitalis purpurea) by chimeric foreign genes and production of cardioactive glycosides. Plant Cell Rep. 9, 121–124
Slightom, J.L., Durand-Tardif, M., Jouanin, L., Tepfer, D. (1986) Nucleotide sequence analysis of TL-DNA of Agrobacterium rhizogenes agropine type plasmid. J. Biol. Chem. 261, 108–121
Stanisich, V.A., Bennett, P.M., Richmond, M.H. (1977) Characterization of a translocating unit encoding resistance to mercuric ions that occurs on a non conjugative plasmid in Pseudomonas aeruginosa. J. Bacteriol. 129, 1227–1233
Sugaya, S., Hayakawa, K., Handa, T., Uchimiya, H. (1989) Cell-specific expression of the rolC gene of the TL-DNA of Ri plasmids in transgenic tobacco plants. Plant Cell Physiol. 30, 649–653
Teeri, T.H., Lehvaslaiho, H., Franck, M., Uotila, J., Heino, P., Palva, E.T., Van Montagu, M., Herrera-Estrella, L. (1989) Gene fusions to lacZ reveal new expression patterns of chimeric genes in transgenic plants. EMBO J. 8, 343–350
Tepfer, D. (1984) Transformation of several species of higher plants by Agrobacterium rhizogenes: Sexual transmission of the transformed genotype and phenotype. Cell 37, 959–967
Vaeck, M., Reynaerts, A., Höfte, H., Jansens, S., De Beuckeleer, M., Dean, C., Zabeau, M., Van Montagu, M., Leemans, J. (1987) Transgenic plants protected from insect attack. Nature 328, 33–37
Velten, J., Schell, J. (1985) Selection-expression plasmid vectors for use in genetic transformation of higher plants. Nucleic Acids Res. 13, 6981–6998
Velten, J., Velten, L., Hain, R., Schell, J. (1984) Isolation of a dual plant promoter fragment from the Ti plasmid of Agrobacterium tumefaciens. EMBO J. 3, 2723–2730
Wagne, H.L., Leopold, A.C. (1969) Ethylene evolution from 2-chloroethyl phosphonic acid. Plant Physiol. 44, 156–158
Weising, K., Schell, J., Kahl, G. (1988) Foreign genes in plants: Transfer, structure, expression, and applications. Annu. Rev. Genet. 22, 421–477
Zambryski, P., Tempé, J., Schell, J. (1989) Transfer and function of T-DNA genes from Agrobacterium Ti and Ri plasmids in plants. Cell 56, 193–201
Author information
Authors and Affiliations
Additional information
We wish to thank Drs. J. Leemans and J. Van Rompaey (Plant Genetic Systems, Gent, Belgium) for providing pGSGluc1; Dr. A. Caplan (Laboratorium voor Genetica, Rijksuniversiteit Gent) for critical reading of the manuscript; Dr. L. Jouanin (INRA, Versailles, france) for a gift of pLJ1. Claforan™ was from Hoechst Japan (Tokyo, Japan). This research was supported, in part, by Grants-in-Aid from the Ministry of Education, Science and Culture, Japan, Special Cooperation Funds for Promoting Science and Technology Agency, Japan, and the Ministry of Health and Welfare, Science Research Fund Subsidy granted to the Japan Health Sciences Foundation. M.Y. is supported by JSPS Fellowships for Japanese Junior Scientists.
Rights and permissions
About this article
Cite this article
Saito, K., Yamazaki, M., Kaneko, H. et al. Tissue-specific and stress-enhancing expression of the TR promoter for mannopine synthase in transgenic medicinal plants. Planta 184, 40–46 (1991). https://doi.org/10.1007/BF00208234
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00208234