Summary
-
1.
The catecholamines dopamine, epinephrine and norepinephrine were detected in alumina extracts of Limulus midgut tissue using high performance liquid chromatography with electrochemical detection. Moderate levels of norepinephrine (28.2±2.1 ng/g) and dopamine (24.0±5.2 ng/g) were detected in the midgut, while epinephrine levels (7.4±0.9 ng/g) were less. Catecholamines were present in all regions along the longitudinal axis of the midgut, and norepinephrine and dopamine levels were highest in posterior regions.
-
2.
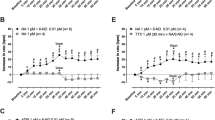
Catecholamines decreased muscle tonus and inhibited spontaneous contractions of the Limulus midgut. Dopamine typically decreased spontaneous midgut activity at doses of 10−8 M or greater, and produced inhibitory actions on all regions of the Limulus midgut. In some preparations epinephrine and norepinephrine elicited a secondary rhythmicity. The actions of dopamine opposed the excitatory effects produced by either proctolin or octopamine.
-
3.
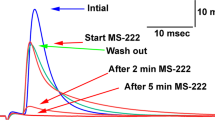
Catecholamines significantly elevated levels of cyclic AMP in Limulus midgut muscle rings. Dopamine (10−5 M) increased cyclic AMP with a time course consistent with its physiological effects. Forskolin and several methyl xanthines increased Limulus midgut cyclic AMP levels and mimicked the inhibitory effects of dopamine on the isolated midgut preparation. Cyclic nucleotide analogues also produced dopamine-like effects on the isolated midgut preparation. Inhibition of cyclic nucleotide phosphodiesterase prior to addition of dopamine enhanced the effect of this amine to decrease baseline muscle tension.
-
4.
The inhibitory effects of 10−5 M dopamine on the midgut persisted in solutions of zero sodium and in the presence of tetrodotoxin. Zero calcium solutions gradually reduced spontaneous midgut activity and the effects of dopamine. Calcium channel blockers did not prohibit dopamine-induced relaxation. These findings suggest that direct, inhibitory actions of dopamine on the Limulus midgut are mediated by increased levels of cyclic AMP.
Similar content being viewed by others
Abbreviations
- cAMP :
-
cyclic adenosine 3′-5′-monophosphate
- CNS :
-
central nervous system
- DA :
-
dopamine
- EPI :
-
epinephrine
- HPLC-EC :
-
high performance liquid chromatography with electrochemical detection
- IBMX :
-
3-isobutyl 1-methyl xanthine
- NE :
-
norepinephrine
- OCT :
-
octopamine
- RIA :
-
radioimmunoassay
References
Anton AH, Sayre DF (1962) A study of the factors affecting the alumina oxide-trihydroxy indole procedure for the analysis of catecholamines. J Pharmacol Exp Ther 138:360–375
Beltz BS, Kravitz EA (1986) Aminergic and peptidergic modulation in Crustacea. J Exp Biol 124:115–141
Cook BJ, Wagner RM (1990) Isolation and chemical characterization of cockroach neuropeptides: the myotropic and hyperglycemic peptides. In: Huber I, Masler EP, Rao BR (eds) Cockroaches as models for neurobiology: applications in biomedical research, vol. 2. CRC Press, Boca Raton Florida, pp 53–83
Cook BJ, Eraker J, Anderson GR (1969) The effect of various biogenic amines on the activity of the foregut of the cockroach, Blaberus giganteus. J Insect Physiol 15:445–455
Elekes K, Florey E, Cahill MA (1988) Morphology, synaptic connections and neurotransmitters of the efferent neurons of the crayfish hindgut. In: Salanki J, S-Rosza K (eds) Neurobiology of invertebrates, vol. 36. Akadémiai Kiadó, Budapest, Hungary, pp 129–146
Elofsson R, Kauri T, Nielsen SO, Stromberg JO (1968) Catecholamine-containing nerve fibers in the hindgut of the crayfish Astacus astacus L. Experientia 24:1159–1160
Florey E (1954) Über die Wirkung von Acetylcholin, Adrenalin, Nor-adrenalin, Faktor I und anderen Substanzen auf den isolierten Enddarm des Fluβkrebses Procambarus clarkii Girard. Z Vergl Physiol 36:1–8
Gies A (1986) Serotonin and dopamine as regulators of adenylate cyclase and relaxation in a smooth muscle of the mussel Mytilus edulis. Comp Biochem Physiol 84C (1): 61–66
Groome JR, Watson WH III (1987) Mechanism for amine modulation of the neurogenic Limulus heart: evidence for involvement of cAMP. J Neurobiol 18 (5): 417–431
Groome JR, Hunt, DF, Townley MA, Tillinghast EK, Vetrovs A, Griffin PR, Alexander JE, Shabonowitz J, Watson WH III (1990) Identification of proctolin in the central nervous system of the horseshoe crab, Limulus polyphemus Peptides 11:205–211
Groome JR, de Tschaschell M, Watson WH III (1992) Peptidergic regulation of the Limulus midgut. J Comp Physiol A
Hartshone DJ (1987) Biochemistry of the contractile process in smooth muscle. In: Johnson LR (ed) Physiology of the gastrointestinal tract, vol 2. Raven Press, New York, pp 423–482
Holman GM, Cook BJ (1970) Pharmacological properties of excitatory neuromuscular transmission in the hindgut of the cockroach, Leucophaea maderae. J Insect Physiol 16:1891–1907
Huddart H (1985) Visceral muscle. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry and pharmacology, vol 11. Pergamon Press, Oxford, pp 131–194
Huddart H (1986) Influence of glutamate, dopamine and 5-hydroxytryptamine on calcium efflux from locust foregut and hindgut visceral muscle. Dependence upon extracellular calcium and calcium channel activity. Comp Biochem Physiol 84C (2):401–408
Huddart H, Oldfield AC (1982) Spontaneous activity of foregut and hindgut visceral muscle of the locust, Locusta migratoria. II. The effect of biogenic amines. Comp Biochem Physiol 73C:303–311
Kamm KE, Stull JT (1989) Regulation of smooth muscle contractile elements by second messengers. Annu Rev Physiol 51:299–313
Klemm N (1972) Monoamine-containing nervous fibres in foregut and salivary gland of the desert locust Schistocerca gregaria Forskal (Orthoptera, Acrididae). Comp Biochem Physiol 43A:207–212
Lange A, Orchard I (1986) Identified octopaminergic neurons modulate contractions of locust visceral muscle via adenosine 3′,5′- monophosphate (cyclic AMP). Brain Res 363:340–349
Lingle C (1981) The modulatory action of dopamine on crustacean foregut neuromuscular preparations. J Exp Biol 94:285–299
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mercer AR, McGregor DD (1982) Neural regulation and pharmacology of the gut of Chione stutchburyi, a bivalve mollusc. Comp Biochem Physiol 73C:243–251
O'Connor EF, Watson WH, Wyse GA (1982) Identification and localization of catecholamines in the nervous system of Limulus polyphemus. J Neurobiol 13 (1):49–60
Orchard I, Belanger JH, Lange AB (1989) Proctolin: a review with emphasis on insects. J Neurobiol 20 (5):470–496
Torre JC de la, Surgeon JW (1976) A methodological approach to rapid and sensitive monoamine histofluorescence using a modified glyoxylic acid technique, the SPG method. Histochemistry 49:81–93
Van Breemen C, Saida K (1989) Cellular mechanisms regulating [Ca2+]in smooth muscle. Annu Rev Physiol 51:315–329
Watson WH III, Groome JR (1989) Modulation of the Limulus heart. Am Zool 29:1287–1303
Wright MS, Cook BJ, Holman GM (1986) Adenylate cyclase in the insect Leucophaea maderae: biochemical properties of the enzyme from hindgut tissue. Comp Biochem Physiol 85B (2):413–418
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Groome, J.R., Lent, C.M. Inhibitory actions of dopamine on Limulus visceral muscle involve a cyclic AMP-dependent mechanism. J Comp Physiol A 170, 787–795 (1992). https://doi.org/10.1007/BF00198989
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00198989