Abstract
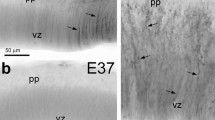
Secretory glial cells in the roof of the last diencephalic prosomer, ependymocytes and hypendymocytes, form the subcommissural organ. The cells of this complex were labelled immunocytochemically, using an antiserum against their specific secretory products. The study aims at the characterization of this cell type in the rat as an anatomical model situation. Radially oriented secretory glial cells remain after birth behind the posterior commissure in the mesencephalic aqueduct. At about postnatal day 10, the cell bodies descend into the conventional ependyma and at postnatal day 25 they assume a compact, rounded appearance. The secretory product they release is involved in the formation of Reissner's fiber. This differentiation in phenotype is not accompanied by a change of the intermediate filament expression. In the adult rat these cells had been labelled immunopositive for cytokeratins 8 and 18 as well as vimentin but not for glial fibrillary acidic protein. DiI-marking from the third ventricle and from the dorsal surface of the brain shows that the basal processes of ependymocytes and hypendymocytes project to the external and internal glial limiting membrane, respectively, through the commissural fiber bundles. Also the subependymal located hypendymocytes have apical processes with contacts to the cerebrospinal fluid. When this secretory cell population is studied with respect to cyto-architectonical changes during ontogeny the results lead to a new understanding of the subcommissural cells. They are not specialized ependymal cells in a regionally restricted and secondary differentiated ependymal area, but rather descendants of an ontogenetically ancient, specific type of radial glia. Characteristic features for all subcommissural cells are that they: (1) appear very early during ontogeny, (2) are derived from a radial oriented glial cell type, (3) carry at least one kinocilium, (4) possess an original intermediate filament pattern, (5) release a secretory product.
Similar content being viewed by others
References
Bouchaud C (1979) Evidence for a multiple innervation of subcommissural ependymocytes in the rat. Neurosci Lett 12:253–258
Castañeyra-Perdomo A, Meyer G, Ferres-Torres R (1883) Development of the subcommissural organ in the albino mouse (a Golgi study). J Hirnforsch 24:363–370
Chouaf L, Didier-Bazes M, Aguera M, Tardy M, Sallanon M, Kitahama K, Belin MF (1989) Comparative marker analysis of the ependymocytes of subcommissural organ in four different mammalian species. Cell Tissue Res 257:255–262
Chouaf L, Didier-Bazes M, Hardin H, Aguera M, Fèvre-Montange M, Voutsinos B, Belin MF (1991) Developmental expression of glial markers in subcommissural organ ependymocytes of the rat: role of the environment. Cell Tissue Res 266:553–561
Didier M, Harandi M, Aguera M, Bancel B, Tardy M, Fages C, Calas A, Stagaard M, Møllgård K, Belin MF (1986) Differential immunocytochemical staining for glial fibrillary acid (GFA) protein, S100 protein and glutamine synthetase in the rat subcommissural organ, nonspecialized ventricular ependyma and adjacent neuropil. Cell Tissue Res 245:343–351
Didier-Bazes M, Aguera M, Chouaf L, Harandi M, Calas A, Meiniel A, Belin MF (1989) Neuronal control of [3H]GABA uptake in the ependymocytes of the subcommissural organ: an in vivo model of neuron-glia interaction. Brain Res 489:137–145
Didier-Bazes M, Couaf L, Hardin H, Aguera M, Fèvre-Montange M, Belin MF (1991) Developmental neuron-glia interaction: role of the serotonin innervation upon the onset of GABA uptake into the ependymocytes of the rat subcommissural organ. Brain Res Dev Brain Res 63:135–139
Fedoroff S (1986) Prenatal ontogenesis of astrocytes. In: Fedoroff S, Vernadakis A (eds) Astrocytes, vol 1. Academic Press, New York, pp 35–74
Gamrani H, Belin MF, Aguera M, Calas A, Pujol JF (1981) Radio-autographic evidence for an innervation of the subcommissural organ by GABA-containing nerve fibres. J Neurocytol 10:411–424
Hilwing I, Gropp A (1972) Staining of constitutive heterochromatin in mammalian chromosomes with a new fluorochrome. Exp Cell Res 75:122–126
Houle J, Fedoroff S (1983) Temporal relationship between the appearence of vimentin and neural tube development. Dev Brain Res 9:189–195
Jackson BW, Grund C, Winter S, Franke WW, Illmensee K (1981) Formation of cytoskeletal elements during mouse embryogenesis. II. Epithelial differentiation and intermediate-sized filaments in early postimplantation embryos. Differentiation 20:203–216
Kasper M (1992) Cytokeratins in intracranial and intraspinal tissue. In: Beck F, Hild W, Kritz W, Pauly JE, Sano Y, Schiebler TH (eds) Advances in anatomy, embryology and cell biology, vol 126. Springer, Berlin Heidelberg New York, pp 17–22
Kasper M, Terpe HJ, Perry G (1990) Age-dependent pattern of intermediate filament protein expression in the human pineal gland. J Hirnforsch 31:215–221
Kiessig H-C (1982) Beiträge zur strukturellen, chemischen und immuncytochemischen Charakterisierung des Komplexes Subcommissuralorgan-Reissnerscher Faden. Dissertation A Karl-Marx-Universität, Leipzig
Kimble JE and Møllgård K (1973) Evidence for basal secretion in the subcommissural organ of the adult rabbit. Z Zellforsch 142:223–239
Köhl W, Linderer T (1973) Zur Entwicklung des Subcommissural-organs der Ratte. Morphologische und histochemische Untersuchungen. Histochemie 33:349–368
Krabbe K (1925) L'organe sous-commissural du cerveau chez les mammiferes. Kungl Dansk Vidensk Sslsk Biol Medd V, 4:1–83
Lehtonen E, Lehto V-P, Vartio T, Badley RA, Virtanen I (1983) Expression of cytokeratin polypeptides in mouse oocytes and preimplantation embryos. Dev Biol 100:158–165
Leonhardt H (1980) Ependym und circumventriculäre Organe. In: Oksche A, Vollrath L (eds) Handbuch der mikroskopischen Anatomie des Menschen, vol 4, part 10. Springer, Berlin Heidelberg New York, pp 177–666
Leonhardt H, Krisch B, Erhardt H (1987) Organization of the neuroglia in the midsagittal plane of the central nervous system: a speculative report. In: Scharrer B, Korf HW, Hartwig HG (eds) Functional morphology of neuroendocrine systems. Springer, Berlin Heidelberg New York, pp 175–187
Lösecke W, Naumann W, Sterba G (1986) Immuno-electron-microscopic analysis of the basal route of secretion in the subcommissural organ of the rabbit. Cell Tissue Res 244:449–456
Marcinkiewicz M, Bouchaud C (1986) Formation and maturation of axo-glandular synapses and concomitant changes in the target cells of the rat subcommissural organ. Biol Cell 56:57–65
Møllgård K, Wiklund L (1979) Serotoninergic synapses on ependymal and hypendymal cells of the rat subcommissural organ. J Neurocytol 8:445–467
Naumann W (1986) (Immunohistochemical investigations on the ontogenesis of the subcommissural organ) Immunhistochemische Untersuchungen zur Ontogenese des Subcommissuralorgans. Acta Histochem [Suppl] 33:265–272
Naumann W, Lehmann W, Debbage P (1993) The subcommissural organ — a secretory glial cell group. In: Oksche A, Rodríguez EM, Fernández-Llebrez P (eds) The subcommissural organ: an ependymal brain gland. Springer, Berlin Heidelberg New York, pp 61–69
Oksche A (1961) Vergleichende Untersuchungen über die sekretorische Aktivität des Subkommissuralorgans und den Gliacharakter seiner Zellen. Z Zellforsch 54:549–612
Olsson R (1958) Studies on the subcommissural organ. Acta Zool (Stockholm) 39:71–102
Olsson R (1969) Phylogeny of the ventricle system. In: Sterba G (ed) Zirkumventrikuläre Organe und Liquor. Fischer, Jena, pp 291–305
Palkovits M (1965) Morphology and function of the subcommissural organ. Stud Biol Acad Sci Hung 4:1–105
Rakic P, Sidman RL (1968) Subcommissural organ and adjacent ependyma: autoradiographic study of their origin in the mouse brain. Am J Anat 122:317–336
Redecker P (1989) Immunohistochemical localisation of glial fibrillary acidic protein (GFAP) and vimentin in the subcommissural organ of the Mongolian gerbil (Meriones unguiculatus). Cell Tissue Res 255:595–600
Rodríguez EM, Oksche A, Hein S, Rodríguez S, Yulis R (1984) Comparative immunocytochemical study of the subcommissural organ. Cell Tissue Res 237:427–441
Rodríguez EM, Rodríguez S, Schoebitz K, Yulis CR, Hoffmann P, Manns V, Oksche A (1989) Light- and electron-microscopic investigation of the rat subcommissural organ grafted under the kidney capsule, with particular reference to immunocytochemistry and lectin histochemistry. Cell Tissue Res 258:499–514
Rodríguez EM, Oksche A, Hein S, Yulis CR (1992) Cell biology of the subcommissural organ. Int Rev Cytol 135:39–121
Schoebitz K, Garrido O, Heinrichs M, Speer L, Rodríguez EM (1986) Ontogenetical development of the chick and duck subcommissural organ and immunocytochemical study. Histochemistry 84:31–40
Schmechel DE, Rakic P (1979) A Golgi study of radial glial cells in developing monkey telencephalon: morphogenesis and transformation into astrocytes. Anat Embryol 156:115–152
Schönwolf GC, Smith JL (1990) Mechanisms of neurulation: traditional viewpoint and recent advances. Development 109:243–270
Stanka P (1963) Über das Subcommissuralorgan bei Schwein und Ratte. Z Mikrosk Anat Forsch 69:395–409
Stieda L (1870) Studien über das zentrale Nervensystem der Wirbeltiere. Z Wiss Zool 20:386–425
Viehweg J, Naumann W, Olsson R (1994) Studies of glial secretion in the nervous system of Asterias rubens (L.). J Brain Res 35:113
Yang H-Y, Lieska N, Shao D, Kriho V, Pappas GD (1993) Immunotyping of radial glia and their glial derivatives during development of the spinal cord. J Neurocytol 22:558–571
Zamboni L, De Martino C (1967) Buffered picric-acid formaldehyde: a new rapid fixation for electron microscopy. J Cell Biol 35:148A
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Ragnar Olsson on the occasion of his 70th birthday. We should also like to thank him for an essential contribution to this paper
Rights and permissions
About this article
Cite this article
Viehweg, J., Naumann, W.W. Radial secretory glia conserved in the postnatal vertebrate brain: a study in the rat. Anat Embryol 194, 355–363 (1996). https://doi.org/10.1007/BF00198537
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00198537