Abstract
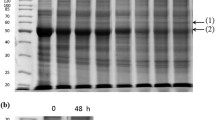
In the green marine alga Dunaliella tertiolecta, a CO2-concentrating mechanism is induced when the cells are grown under low-CO2 conditions (0.03% CO2). To identify proteins induced under low-CO2 conditions the cells were labelled with 35SO4 2−, and seven polypeptides with molecular weights of 45, 47, 49, 55, 60, 68 and 100 kDa were detected. The induction of these polypeptides was observed when cells grown in high CO2 (5% CO2 in air) were switched to low CO2, but only while the cultures were growing in light. Immunoblot analysis of total cell protein against pea chloroplastic carbonic anhydrase polyclonal antibodies showed immunoreactive 30-kDa bands in both high- and low-CO2-grown cells and an aditional 49-kDa band exclusively in low-CO2-grown cells. The 30-kDa protein was shown to be located in the chloroplast. Western blot analysis of the plasmamembrane fraction against corn plasma-membrane AT-Pase polyclonal antibodies showed 60-kDa bands in both high- and low-CO2 cell types as well as an immunoreactive 100-kDa band occurring only in low-CO2-grown cells. These results suggest that there are two distinct forms of both carbonic anhydrase and plasma-membrane ATPase, and that one form of each of them can be regulated by the CO2 concentration.
Similar content being viewed by others
Abbreviations
- CA:
-
carbonic anhydrase
- DIC:
-
dissolved inorganic carbon (CO2+ HCO3 −)
- CCM:
-
CO2-concentrating mechanism
- low CO2 :
-
air containing 0.03% CO2
- high CO2 :
-
air supplemented with 5% CO2 (v/v)
References
Aizawa, K., Miyachi, S. (1984) Carbonic anhydrase located on cell surface increases the affinity for inorganic carbon in photosynthesis of Dunaliella tertiolecta. FEBS Lett. 173, 41–44
Aizawa, K., Miyachi, S. (1986) Carbonic anhydrase and CO2 concentrating mechanisms in microalgae and cyanobacteria. FEMS Micro Rev. 39, 215–233
Badger, M.R. (1987) The CO2 concentrating mechanism in aquatic phototrophs. In: The biochemistry of plants: a comprehensive treatise, vol. 10: Photosynthesis, pp. 219–274, Hatch, M.D., Boardman, N.K., eds. Academic Press, New York
Badger, M.R., Kaplan, A., Berry, J.A. (1980) Internal inorganic carbon pool of Chlamydomonas reinhardtii: evidence for a carbon dioxide concentrating mechanism. Plant Physiol. 66, 407–413
Beardall, J. (1981) CO2 accumulation by Chlorella saccharophila (Chlorophyceae) at low external pH: evidence for the active transport of inorganic carbon at the chloroplast envelope. J. Phycol. 17, 371–373
Beardall, J., Raven, J. (1981) Transport of inorganic carbon and the CO2 concentrating mechanism in Chlorella emersonii (Chlorophyceae). J. Phycol. 17, 134–141
Booth, W.A., Beardall, J. (1991) Effects of salinity on inorganic carbon utilization and carbonic anhydrase activity in the halotolerant alga Dunaliella salina (Chlorophyta). Phycologia 30, 220–225
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of proteindye binding. Anal. Biochem. 72, 248–254
Coleman, J.R., Grossman, A.R. (1984) Biosynthesis of carbonic anhydrase in Chlamydomonas reinhardtii during adaptation to low CO2. Proc. Natl. Acad. Sci. USA 81, 6049–6053
Dionisio-Sese, M.L., Miyachi, S. (1992) The effect of sodium chloride on carbonic anhydrase activity in marine microalgae. J. Phycol. 28, 619–624
Fukuzawa, H., Fujiwara, S., Yamamoto, Y, Dionosio-Sese, M.L., Miy-achi, S. (1990) cDNA cloning, sequence, and expression of carbonic anhydrase in Chlamydomonas reinhardtii: Regulation by environmental CO2 concentration. Proc. Natl. Acad. Sci. USA, 87, 4383–4387
Gehl, K.A., Colman, B., Sposato, L.M. (1990) Mechanism of inorganic carbon uptake in Chlorella saccharophila: the lack of involvement of carbonic anhydrase. J. Exp. Bot. 41, 1385–1391
Geraghty, A.M., Anderson, J.C., Spalding, M.H. (1990) A 36 kDa kilodalton limiting-CO2 induced polypeptide of Chlamydomonas is distinct from 37 kilodalton periplasmic carbonic anhydrase. Plant Physiol. 93, 116–121
Gómez-Pinchetti, J.L., Ramazanov, Z., Fontes, A., García Reina, G. (1992) Photosynthetic characteristics of Dunaliella salina (Chlorophyceae, Dunaliellales) in relation to β-carotene content. J. Appl. Phycol. 4, 11–15
Goyal, A., Tolbert, N.E. (1989) Uptake of inorganic carbon by isolated chloroplasts from air-adapted Dunaliella. Plant Physiol. 89, 1264–1269
Goyal, A., Shiraiwa, Y, Husic, D.H., Tolbert, N.E. (1992) External and internal carbonic anhydrase in Dunaliella species. Mar. Biol. 113, 349–355
Husic, H.D., Kitayama, M., Togasaki, R.K., Moroney, J.V., Morris, K.L., Tolbert, N.E. (1989) Identification of intracellular carbonic anhydrase in Chlamydomonas reinhardtii which is distinct from the periplasmic form of the enzyme. Plant Physiol. 89, 904–909
Jiménez del Río, M., Ramazanov, Z., García-Reina, G. (1994) Dark induction of nitrate reductase in the halophylic alga Dunaiiella salina. Planta 192, 40–45
Karlsson, J., Ramazanov, Z., Hiltonnen, TH., Gardestrom, P., Sa-muelsson, G. (1994) Effect of vanadate on photosynthesis and the ATP/ADP ratio in Chlamydomonas reinhardtii. Planta 192, 46–51
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685
Larsson, C. (1985) Plasma membranes. In: Modern methods of plant analysis, vol. 1, pp. 85–104, Linskens, H.F., Jackson, J.F., eds. Springer-Verlag, Berlin
Manuel, L.J., Moroney, J.V. (1988) Inorganic carbon accumulation in Chlamydomonas reinhardtii: New proteins are made during adaption to low CO2. Plant Physiol. 88, 491–496
Marcus, Y, Volokita, M., Kaplan, A. (1984) The location of the transporting system for inorganic carbon and the nature of the form translocated in Chlamydomonas reinhardtii. J. Exp. Bot. 35, 1136–1144
Martinez, F., Villarejo, A., Ramazanov, Z., Orus, M.I. (1992) Carbonic anhydrase independent stimulation of inorganic carbon fixation mediated by glucose. In: Research in photosynthesis, vol. 3, pp. 803–806, Murata, N., ed. Kluwer Academic Publishers, The Netherlands
Moroney, J.V., Mason, C.B. (1991) The role of the chloroplast in inorganic carbon acquisition by Chlamydomonas reinhardtii. Can. J. Bot. 69, 1017–1024
Pick, U. (1992) ATPases and ion transport in Dunaliella. In: Dunaliella: Physiology, biochemistry and biotechnology, pp. 63–97, Avron, M., Ben-Amotz, A., eds. CRC Press, Boca Raton, Florida, USA
Pronina, N.A., Ramazanov, Z., Semenenko, V.E. (1981) Carbonic anhydrase activity of Chlorella cells as a function of CO2 concentration. Fiziol. Rast. [In Russ.] 28, 494–502
Ramazanov, Z., Cardenas, J. (1992) Inorganic carbon transport across cell compartments of the halotolerant alga Dunaliella salina. Physiol. Plant. 85, 121–128
Ramazanov, Z., Mason, C., Geraghty, A., Spalding, M., Moroney, J. (1993) The low CO2-inducible 36-kilodalton protein is localized to the chloroplast envelope of Chlamydomonas reinhardtii. Plant Physiol. 101, 1195–1999
Raven, J.A., Lucas, W.J. (1985) The energetics of carbon acquisition. In: Inorganic carbon uptake by aquatic photosynthetic organisms, pp. 305–324, Lucas, W.J., Berry, J.A., eds. American Society of Plant Physiologists, Rockville, Mo., USA
Rawat, M., Moroney, J.V. (1991) Partial characterization of a new isoenzyme of carbonic anhydrase isolated from Chlamydomonas reinhardtii. J. Biol. Chem. 266, 9719–9723
Rotatore, C., Roger, R.L., Colman, B. (1992) Active uptake of CO2 during photosynthesis in the green alga Eremosphaera viridis is mediated by a CO2-ATPase. Planta 188, 539–545
Samuelsson, G., Palmqvist, K., Ramazanov, Z., Sundblad, L-G., (1990) Photosynthetic acclimation to low carbon concentrations in Chamydomonas reinhardtii. In: Current research in photosynthesis, vol. 4, pp. 18441–18448, Baltscheffsky, M., ed. Kluwer Academic Publishers, The Netherlands
Spalding, M.H., Spreitzer, R.J., Ogren, W.L. (1983) Carbonic anhydrase deficient mutant of Chlamydomonas reinhardtii requires elevated carbon dioxide concentrations for photoautotrophic growth. Plant Physiol. 73, 268–272
Spalding, M.H., Jeffrey M. (1989) Membrane-associated polypeptides induced in Chlamydomonas reinhardtii by limiting CO2 concentration. Plant Physiol. 89, 133–137
Spalding, M., Winder, T.L., Anderson, J.C., Geraghty, A.M., Marek, L.F. (1991) Changes in protein and gene expression during induction of the CO2-concentrating mechanism in wild-type and mutant Chlamydomonas. Can. J. Bot. 69, 1008–1016
Sültemeyer, D.F., Fock, H.P., Canvin, D.T. (1990) Mass spectrometric measurement of intracellular carbonic anhydrase activity in high and low Ci cells of Chlamydomonas. Studies using 18O exchange with 13C/18O labeled bicarbonate. Plant Physiol. 94, 1250–1257
Sültemeyer, D.F., Schmidt, C., Fock, H.P. (1993) Carbonic anhydrase in higher plants and aquatic microorganisms. Physiol. Plant. 88, 179–190
Thielmann, J., Tolbert, N.E., Goyal, A., Senger, H. (1990) Two systems for concentrating CO2 and bicarbonate during photosynthesis by Scenedesmus. Plant Physiol. 92, 622–629
Thielmann, J., Goyal, A., Tolbert N.E. (1992) Two polypeptides in the inner chloroplast envelope of Dunaliella tertiolecta induced by low CO2. Plant Physiol. 100, 2113–2115
Widell, S. (1987) Purification of plasmalemma with aqueous polymer two-phase partition. Physiol. Plant. 69, 727–730
Wintermans, J.F.G., De Mots, A. (1965) Spectrophotometric characteristics of chlorophyll a and b and their pheophytins in ethanol. Biochim. Biophys. Acta 109, 448–453
Zenvirth, D., Kaplan, A. (1981) Uptake and efflux of inorganic carbon in Dunaliella salina. Planta 152, 8–12
Author information
Authors and Affiliations
Additional information
We thank Prof. John Coleman for providing antibodies raised against pea chloroplast CA, Dr. James V. Moroney for providing antibodies raised against the 37-kDa periplasmic carbonic anhydrase of CO2 Chlamydomonas reinhardtii, and Prof. Leonard T. Robert for a gift of corn plasma-membrane 100-kDa ATPase antibodies. We thank Dr. Jeanine Olsen (University of Groningen, the Netherlands) for style comments. This work was supported by the Institute Tecnológico de Canarias (Spain).
Rights and permissions
About this article
Cite this article
Ramazanov, Z., Sosa, P.A., Henk, M.C. et al. Low-CO2-inducible protein synthesis in the green alga Dunaliella tertiolecta . Planta 195, 519–524 (1995). https://doi.org/10.1007/BF00195709
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00195709