Abstract
There is an increased accumulation of message for the catalytic (70-kDa) subunit of the tonoplast H+-ATPase in leaves of tomato (Lycopersicon esculentum L.) plants responding to NaCl. To determine if abscisic acid (ABA) mediates this response, message accumulation was examined in treatments designed to separate exposure to NaCl from increases in endogenous ABA. Under three different experimental conditions, salt-induced changes in the accumulation of 70-kDa message were unrelated to any change in endogenous ABA. The results were as follows: (i) under drought stress, plants accumulated levels of ABA similar to those measured in salt-treated plants; however, no increase in 70-kDa subunit message was observed; (ii) the ABA-deficient mutant sitiens exhibited an increased accumulation of message despite the absence of NaCl-induced accumulation of ABA; and (iii) the inhibitor of general isoprenoid biosynthesis, Lovastatin, blocked NaCl-induced accumulation of ABA but did not alter NaCl-induced accumulation of message. In addition to these three experimental responses, application of exogenous ABA increased endogenous ABA levels without any comparable increase in message accumulation. Based on these results, it is concluded that ABA does not mediate the NaCl-induced accumulation of 70-kDa subunit tonoplast H+ -ATPase message accumulation in tomato.
Similar content being viewed by others
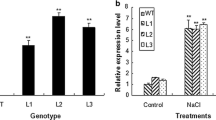
Abbreviations
- ABA:
-
abscisic acid
- WT:
-
wild type
References
Binzel ML (1994) Alterations in H+-ATPase gene expression in response to salt. In: Cherry JH (ed) Molecular and cellular mechanisms of stress tolerance in plants (NATO ASI Series vol H86), Springer, Berlin Heidelburg New York, pp 429–442
Binzel ML (1995) NaCl-induced accumulation of tonoplast and plasma membrane H+-ATPase message in tomato. Physiol Plant 94: in press
Binzel ML, Hess FD, Bressan RA, Hasegawa PM (1988) Intracellular compartmentation of ions in salt-adapted tobacco cells. Plant Physiol 86: 607–614
Blumwald E, Poole RJ (1987) Salt tolerance in suspension cultures of sugar beet: Induction of Na+/H+ antiport activity at the tonoplast by growth in salt. Plant Physiol 83: 884–887
Bostock RM, Quatrano RS (1992) Regulation of Em gene expression in rice.Interaction between osmotic stress and abscisic acid. Plant Physiol 98: 1356–1363
Bremberger C, Haschke H-P, Lüttge U (1988) Separation and purification of the tonoplast ATPase and pyrophosphatase from plants with inducible Crassulacean acid metabolism. Planta 175: 465–470
Bremberger C, Lüttge U (1992) Dynamics of tonoplast proton pumps and other tonoplast proteins of Mesembryanthemum crystallinum L. during the induction of Crassulacean acid metabolism. Planta 188: 575–580
Chen THH, Gusta LV (1983) Abscisic acid-induced freezing resistance in cultured plant cells. Plant Physiol 73: 71–75
Chandler PM, Robertson M (1994) Gene expression regulated by abscisic acid and its relation to stress tolerance. Annu Rev Plant Physiol Plant Mol Biol 45: 113–141
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159
Cohen A, Plant AL, Moses S, Bray EA (1991) Organ-specific and environmentally regulated expression of two abscisic acid-induced genes of tomato. Nucleotide sequence and analysis of the corresponding cDNAs. Plant Physiol 97: 1367–1374
Cohen JD (1986) 13C6[Benzene ring]-indole-3-acetic acid: A new internal standard for quantitative mass spectral analysis of indole-3-acetic acid. Plant Physiol 80: 14–19
Dunlap JR, Robacker KM (1990) Abscisic acid alters the metabolism of indole-3-acetic acid in senescing flowers of Cucumis melo L. Plant Physiol 94: 870–874
Dupont FM (1992) Salt-induced changes in ion transport: regulation of primary pumps and secondary transporters. In: Cooke DT, Clarkson DT (eds) Transport and receptor proteins of plant membranes. Plenum Press, New York, pp 91–100
Epstein E (1972) Mineral nutrition of plants: principles and perspectives. John Wiley and Sons. New York, pp 38–39
Guerrero FD, Jones JT, Mullet JE (1990) Turgor-responsive gene transcription and RNA levels increase rapidly when pea shoots are wilted. Sequence and expression of three inducible genes. Plant Mol Biol 15: 11–26
Guiltinan MJ, Marcotte WR, Quatrano RS (1990) A plant leucine zipper protein that recognizes an abscisic acid response element. Science 250: 267–271
LaRosa PC, Hasegawa PM, Rhodes D, Clithero JM, Watad AA, Bressan RA (1987) Abscisic acid stimulated osmotic adjustment and its involvement in adaptation of tobacco cells to NaCl. Plant Physiol 85: 174–181
Marcotte WR, Russell SH, Quatrano RS (1989) Abscisic acid responsive sequences from the Em gene of wheat. Plant Cell 1: 969–976
Matsumoto H, Chung GC (1992) Increase in proton-transport activity of tonoplast vesicles as an adaptive response of barley roots to NaCl stress. Plant Cell Physiol 29: 1133–1140
Mundy J, Chua N-H (1988) Abscisic acid and water-stress induce the expression of a novel rice gene. EMBO J 7: 2279–2286
Narasimhan ML, Binzel ML, Perez-Prat E, Chen Z, Nelson DE, Singh NK, Bressan RA, Hasegawa PM (1991) NaCl regulation of tonoplast ATPase 70-kD subunit mRNA in tobacco cells. Plant Physiol 97: 562–568
Reuveni M, Bennett AB, Bressan RA, Hasegawa PM (1990) Enhanced H+ transport capacity and ATP hydrolysis activity of the tonoplast H+-ATPase after NaCl adaptation. Plant physiol 94: 524–530.
Roberts DL, Heckman RA, Hege BP, Bellin SA (1968) Synthesis of (RS)-abscisic acid. J Org Chem 33: 3566–3569
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning. A laboratory manual, edn. 2. Cold Spring Harbor Laboratory Press, Cold-Spring Harbor, NY
Skriver K, Mundy J (1990) Gene expression in response to abscisic acid and osmotic stress. Plant Cell 2: 503–512
Struve I, Rausch T, Bernasconi R, Taiz L (1990) Structure and function of the promoter of the carrot V-type H+-ATPase catalytic subunit gene. J Biol Chem 265: 251–262
Tal M, Nevo Y (1973) Abnormal stomatal behavior and root resistance, and hormonal imbalance in three wilty mutants of tomato. Biochem Gen 8: 291–300
Taylor IB, Linforth RST, Al-Nayeb RJ, Bowman WR, Marples BA (1988) The wilty mutants flacca and sitiens are impaired in the oxidation of ABA-aldehyde to ABA. Plant Cell Environ 11: 739–745
Thomas JC, McElwain EF, Bohnert HJ (1992) Convergent induction of osmotic stress-responses. Abscisic acid, cytokinin, and the effects of NaCl. Plant Physiol 100: 416–423
Yamaguchi-Shinozaki K, Koizumi M, Urao S, Shinozaki K (1992) Molecular cloning and characterization of 9 cDNAs for genes that are responsive to desiccation in Arabidopsis thaliana: Sequence analysis of one cDNA clone that encodes a putative transmembrane channel protein. Plant Cell Physiol 33: 217–224
Author information
Authors and Affiliations
Additional information
The generous gift of Lovastatin from Merck is gratefully acknowledged. The authors also thank B. Sorenson and J. Molina for their technical assistance. This research was supported in part by National Research Initiative Competitive Grants Program/U.S. Department of Agriculture award #93-37100-8951. This manuscript reports research conducted by the Texas Agricultural Experiment Station, The Texas A&M University System.
Rights and permissions
About this article
Cite this article
Binzel, M.L., Dunlap, J.R. Abscisic acid does not mediate NaCl-induced accumulation of 70-kDa subunit tonoplast H+ -ATPase message in tomato. Planta 197, 563–568 (1995). https://doi.org/10.1007/BF00191561
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00191561