Summary
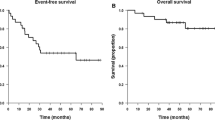
A pilot study was performed combining cis-diamminedichloroplatinum (CDDP) and radiation therapy to treat patients with high-grade astrocytomas. CDDP at a dose of 40 mg/ m2/ week intravenously was given during the course of cranial irradiation. Following irradiation, CDDP was given every three weeks on a schedule of 35–40 mg/ m2/ day for three days until toxicity became unacceptable or until tumor progression occurred. Radiation therapy consisted of 6 000 rads over a seven week period or 5 000 rads followed by an additional 1500 rads to the tumor site. Patients were followed by computerized axial tomography (CT) scan and neurologic examination. Thirty patients were entered onto the study; 22 were considered evaluable. The median survival was 53 weeks and the median time to progression was 21 weeks. Toxicity was generally tolerable; however, ototoxicity may be enhanced by this treatment. CDDP combined with cranial irradiation is tolerable and feasible, although close follow-up is recommended in case CDDP has to be temporarily interrupted.
Similar content being viewed by others
References
Walker MD, Gehan EA: Clinical studies in malignant gliomas and their treatment with the nitrosoureas. Cancer Treat Rep 60:713–716, 1976.
Stewart DJ, Leavens M, Maor M, Feun LG, Luna M, Bonura J, Loo TL, Benjamin RS: Cis-diamminedichloroplatinum: Human central nervous sytem distribution. Cancer Res 42:2474–2479, 1982.
Wolpert DE, Filippes MK: Antitumor activity of cis-diamminedichloroplatinum (I1). Cancer Treat Rep 63:1453–1458, 1979.
Murthy AK, Rossof AH, Anderson KM, Hendreckson FR: Cytotoxicity and influence on radiation dose response curve of cis-diamminedichloroplatinum (II) (CDDP). Int J Rad Oncol Biol Phys 5:1411–1415, 1979.
Dritschilo A, Piro AJ, Kelman AD: The effect of cis-platinum on the repair of radiation damage in plateau phase chinese hamster (V-79) cells. Int J Radiat Oncol Biol Phys 5:1345–1349, 1979.
Double EB, Richmond RC: Platinum complexes as radiosensitizers of hypoxic mammalian cells. Br J Cancer 37 (Suppl III):98–102, 1978.
Leventhal BG, Freeman A: Cis-diamminedichloroplatinum —A phase II study in pediatric malignancies. Proc Am Assoc Cancer Res 20:197, 1979.
Khan A, McCullough, Borts F, Sinks LF: Update on use of cis-platinum in CNS malignancies. Proc Am Soc Clin Oncol 21:390, 1980.
Rozenweig M, Von Hoff DD, Slaik M, Muggia F: Cis-diamminedichloroplatinum: A new anticancer drug. Ann Intern Med 86:803–812, 1977.
Eagan RT, Childs DS, Layton DD, et al: Dianhydrogalactitol and radiation therapy. JAMA 241:2046–2050, 1979.
Weir B, Band P, Urtasun R, et al: Radiotherapy and CCNU in the treatment of high-grade supratentorial astrocytoma. J Neurosurg 45:129–134, 1976.
Walker MD, Green SB, Byar DP, et al: Randomized comparisons of radiotherapy and nitrosoureas for the treatment of malignant glioma after surgery. N Engl J Med 303:1323–1329, 1980.
Reagan TJ, Bisel HF, Childs DS Jr, et al: Controlled study of CCNU and radiation therapy in malignant astrocytoma. J Neurosurg 44:186–190, 1976.
Levin VA, Wilson CB, Davis R, Wara WM, Pisher TL, Irwin L: A phase III comparison of BCNU, hydroxyurea, and radiation therapy to BCNU and radiation therapy for treatment of primary malignant gliomas. J Neurosurg 51:526–532, 1979.
Yung WK, Rankin JL, Shapiro WR: Heterogeneous chemosensitivities of human glioma cell lines by colony forming assay (CFA). Proc Am Soc Clin Oncol 21:217, 1981.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Feun, L.G., Stewart, D.J., Maor, M. et al. A pilot study of cis-diamminedichloroplatinum and radiation therapy in patients with high grade astrocytomas. J Neuro-Oncol 1, 109–113 (1983). https://doi.org/10.1007/BF00182955
Issue Date:
DOI: https://doi.org/10.1007/BF00182955