Summary
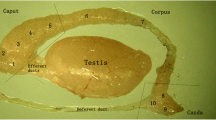
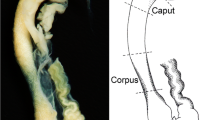
The distribution of protein gene product 9.5 (PGP) and ubiquitin in the spermatozoa and epithelial cells in the different regions of the rat duetus epididymidis (proximal caput, distal caput, corpus and cauda) was studied by Western blotting analyses and electron microscopical immunogold labelling. Western blotting analyses showed that the PGP immunoreactive band was very intense in the caput and cauda epididymidis and almost irrelevant in the corpus, while the ubiquitin immunoreactive band was intense in the distal caput and cauda. No ubiquitin immunoreactive band was observed in the proximal caput and only a very weak band was seen in the corpus. The results of electron microscopical immunogold labelling varied from one epididymal region to another. The proximal caput epididymidis presented immunoreaction to PGP in the rough endoplasmic reticulum, cytosol, mitochondria and microvilli of most principal cells, and in the cytosol, rough endoplasmic reticulum and mitochondria of most basal cells. No ubiquitin immunoreaction was observed in this epididymal region. In the distal caput epididymidis, PGP immunoreactivity was detected in some principal and basal cells in the same intracellular locations as described in the proximal caput. In this region, ubiquitin immunoreactivity appears in the apical cytosol and mitochondria of principal cells. The corpus epididymidis showed no immunoreaction to PGP or ubiquitin. In the cauda epididymidis, immunostaining to PGP was observed in most clear cells and in isolated principal cells. The intracellular location of PGP in both cell types was the cytosol, mitochondria and microvilli. Ubiquitin immunoreactivity was detected in the perinuclear cytosol and mitochondria — but not in the digestive vacuoles — of some clear cells. Scanty ubiquitin immunolabelling was also found in the microvilli, cytosol and mitochondria of some principal cells. The head of the spermatozoa present in the ductal lumen in all epididymal regions immunoreacted intensely to PGP. Ubiquitin was detected in the intermediate piece and residual cytoplasm of intraluminal spermatozoa present in the corpus and cauda epididymidis. These findings suggest that a non-ubiquitinated PGP irnrnunoreactive protein is secreted by the principal cells in caput epididymidis and binds the spermatozoon heads. It is possible that the clear cells of the cauda epididymidis secrete the ubiquitin that binds to spermatozoon tail.
Similar content being viewed by others
References
Agell, N. & Mezquita, C. (1988) Cellular content of ubiquitin and formation of ubiquitin conjugates during chicken spermatogenesis. Biochem. J. 250, 883–9.
Anderson, V. E. R., Hajimohammadreza, I., Gallo, J. M., Anderson, B. H., Uney, J., Brown, A. W., Nolan, C. C., Cavanagh, J. B. & Leigh, P. N. (1992) Ubiquitin, PGP 9.5 and dense body formation in trimethyltin intoxication: differential neuronal responses to chemically induced cell damage. Neuropathol. Appl. Neurobiol. 1, 360–75.
Cooper, T. G. (1990) Secretory proteins from the epididymis and their clinical relevance. Andrologia (suppl) 22, 155–65.
Crooke, S. T., Mirabelli, C. K., Ecker, D. J., Butt, T. R., Jonnalagadda, S., Dixon, S., Müller, L., Brown, F., Weber, P. & Monia, B. P. (1990) Studies on the structure and function of ubiquitin. In Protein Design and the Development of New Therapeutics and Vaccines (edited by Hook, J. B. & Poste, G.), pp. 425–45. New York: Plenum Press.
Doran, J. F., Jackson, P., Kynoch, P. A. M. & Thompson, R. J. (1983) Isolation of PGP 9.5, a new human neuron-specific protein detected by high resolution of two-dimensional electrophoresis. J. Neurochem. 40, 1542–7.
Francavilla, S., DeMartino, C., Barcelona, P. S. & Natali, P. G. (1983) Ultrastructural and immunohistochemical studies of rat epididymis. Cell Tissue Res. 233, 523–37.
Fried, V., Smith, H. T., Hildebrandt, E. & Weiner, K. (1987) Ubiquitin has intrinsic proteolytic activity: implication for cellular regulation. Proc. Natl Acad. Sci. USA 84, 3685–9.
Goldstein, G., Scheid, M., Hammerling, U., Boyse, E. A., Schlesinger, D. H. & Niall, H. D. (1975) Isolation of a polypeptide that has lymphocyte-differentiating properties and is probably represented universally in living cells. Proc. Natl Acad. Sci. USA 72, 11–5.
Gulbenkian, S., Wharton, J. & Polar, J. M. (1987) The visualisation of cardiovascular innervation in the guinea pig using an antiserum to protein gene product 9.5 (PGP 9.5). J. Autonom. Nerv. Syst. 18, 235–47.
Hoffer, A. P., Hamilton, D. W. & Fawcett, D. W. (1973) The ultrastructure of the principal cells and intraepithelial leukocytes in the initial segment of the rat epididymis. Anat. Rec. 175, 169–202.
Jackson, P., Thompson, V. M. & Thompson, R. J. (1985) A comparison of the evolutionary distribution of the two neuroendocrine markers, neuron-specific enolase and protein gene product 9.5. J. Neurochem. 45, 185–90.
Kent, C. & Clarke, P. J. (1991) The immunolocalisation of the neuroendocrine specific protein PGP 9.5 during neurogenesis in the rat. Dev. Brain Res. 58, 147–50.
Kent, C. & Rowe, H. L. (1992) The immunolocalisation of ubiquitin carboxyl-terminal hydrolase (PGP 9.5) in developing paraneurons in the rat. Dev. Brain Res. 68, 241–6.
Laemmli, U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–5.
Laszlo, L., Doherty, F. J., Watson, A., Self, T., Landon, N., Lowe, J. & Mayer, J. (1991) Immunogold localization of ubiquitin-protein conjugates in primary (azurophilic) granules of polymorphonuclear neutrophils. FEBS Lett. 279, 175–8.
Lippert, T. H., Seeger, H., Schieferstein, G. & Voelter, W. (1993) Immunoreactive ubiquitin in human seminal plasma. J. Androl. 14, 130–1.
Lowe, J., Blanchard, A., Morrell, K., Lennox, G., Reynolds, L., Billett, M., Landon, M. & Mayer, J. (1988) Ubiquitin is a common factor in intermediate filament inclusion bodies of diverse type in man including those of Parkinson's disease, Pick's disease, and ALzheimer's disease, as well as Rosenthal fibres in cerebellar astrocytomas, cytoplasmic bodies in muscle, and Mallory bodies in alcoholic liver disease. J. Pathol. 155, 9–15.
Manetto, V., Abdul-karim, F. W., Pery, G., Tabton, M. & Autilio-gambetti, P. (1989) Selective presence of ubiquitin in intracellular inclusions. Am. J. Pathol. 134, 505–13.
Mayer, R. J., Lowe, J., Landon, M., Mcdermott, H., Tuckwell, J., Doherty, F. & Laszlo, L. (1991) Ubiquitin and lysosome system: molecular immunopathology reveals connection. Biomed. Biochim. Acta 50, 4–6.
Moore, H. D. M. & Bedford, J. M. (1979) The differential absorptive activity of epithelial cells of the rat epididymis before and after castration. Anat. Rec. 193, 313–28.
Murti, K. G., Smith, H. T. & Fried, V. A. (1988) Ubiquitin is a component of the microtubule network. Proc. Natl Acad. Sci. USA 85, 3019–23.
Ohta, M., Marceau, N., Perry, G., Manetto, V., Gambetti, P., Autilio-gambetti, L., Metuzls, J., Kawahara, H., Cadrin, M. & French, S. W. (1988) Ubiquitin is present in cytokeratin intermediate filaments and Mallory bodies of hepatocytes. Lab. Invest. 59, 848–56.
Properzi, G., Cordeschi, G. & Francavilla, S. (1992) Post-natal development and distribution of peptide containing nerves in the genital system of the male rat. An immunohistochemical study. Histochemistry 97, 61–9.
Rode, J., Dhillon, A. P., Doran, J. J., Jackson, P. & Thompson, R. J. (1985) PGP 9.5 a new marker for human neuroendocrine tumors. Histopathology 9, 147–58.
Santamaría, L., Martín, R., Paniagua, R., Fraile, B., Nistal, M., Terenghi, G. & Polak, J. M. (1993) Protein gene product 9.5 and ubiquitin immunoreactivities in rat epididymis epithelium. Histochemistry 100, 131–8.
Schwartz, A. L., Ciechanover, A., Brandt, R. A. & Greuze, R. J. (1988) Immunoelectron microscopic localization of ubiquitin in hepatoma cells. EMBO J. 7, 2961–6.
Towbin, H., Staehlin, T. & Gordon, J. (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl Acad. Sci. USA 76, 4350–4.
Tuner, T. T. (1991) Spermatozoa are exposed to a complex microenvironment as they traverse the epididymis. In The Male Germ Cell. Spermatogonium to Fertilization. (edited by Robarie, E.), pp. 364–87. New York: Nyas.
Wilkinson, K. D., Lee, K., Deshpande, S., Duerken-Hughes, P., Boss, J. M. & Pohl, J. (1989) The neuronspecific protein PGP 9.5 is a ubiquitin carboxyl-terminal hydrolase. Science 246, 670–3.
Wilkinson, K. D., Deshpande, S. & Larsen, C. N. (1992) Comparisons of neuronal (PGP 9.5) and non-neuronal ubiquitin C-terminal hydrolases. Biochem. Soc. Trans. 20, 631–7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martín, R., Santamaría, L., Fraile, B. et al. Ultrastructural localization of PGP 9.5 and ubiquitin immunoreactivities in rat ductus epididymidis epithelium. Histochem J 27, 431–439 (1995). https://doi.org/10.1007/BF00173708
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00173708