Summary
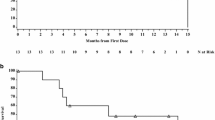
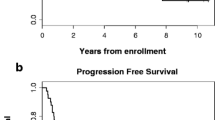
Twenty-four patients with advanced epidermoid carcinoma of the esophagus were treated with trimetrexate (TMTX), a lipid soluble non-classical antifol. Patients were given TMTX 8 mg/m2 intravenously day 1–5 every 28 days. In nine of these patients the dose was escalated to 12 mg/m2 day 1–5 every 28 days. Three patients had a partial response (95% confidence limit 3–32%) with a median response duration of 14 weeks. No hematologic toxicity was documented. Two patients developed moderate stomatitis and only 3 patients experienced any nausea or vomiting. The median survival of all patients is 12 weeks. It is concluded that a higher dose of TMTX should be studied in patients with esophageal cancer in order to assess the true therapeutic value of the agent at a dose closer to the median tolerated dose. A phase II ECOG study using TMTX 12 mg/m2 intravenously day 1–5 every 21 days is currently being conducted.
Similar content being viewed by others
References
Earlam R, Cunha-Melo JR: Oesophageal squamous cell carcinoma: I. A critical review of surgery. Br J Surg 67:381–390, 1980
Earlam R, Cunha-Melo JR: Oesophageal squamous cell carcinoma: II. A critical review of radiotherapy. Br J Surg 67:457–461, 1980
Kolaric K, Mqricic Z, Dujmovic I, Roth A: Therapy of advanced esophageal cancer with bleomycin, irradiation and combination bleomycin and irradiation. Tumori 62:255–262, 1976
Engstrom P, Lavin P, Kalssen D: Phase II evaluation of mitomycin and cisplatin in advanced esophageal carcinoma. Cancer Treat Rep 67:713–715, 1983
Ezdinli E, Gelber R, Desai D, Falkson G, Moertel CG, Hahn RG: Chemotherapy of advanced esophageal carcinoma: Eastern Cooperative Oncology Group experience. Cancer 46:2149–2153, 1980
Moertel C, Schutt A, Reitemeier R, Hahn RG: Therapy for gastrointestinal cancer with the nitrosoureas alone in drug combination. Cancer Treat Rep 60:729–732, 1976
Falkson G: Methyl-GAG (NSC 32946) in the treatment of esophageal cancer. Cancer Chemother Rep 55:209–212, 1971
Kelsen DP, Bains MS, Cvitkovic E, Golbey R: Vindesine in the treatment of esophageal carcinoma: A phase II study. Cancer Treat Rep 63:2019–2021, 1979
Coonley C, Bains M, Heelan M, Dukeman M, Kelsen DP: VP-16–213 in the treatment of esophagel cancer: A Phase II trial. Cancer Treat Rep 67:397–398, 1983
Kelsen D, Nanus D, Lipperman R, Eisenberger M: Phase II trial of Ifosfamide (Ifos) in epidermoid carcinoma of the esophagus (ECE): Unexpectedly severe toxicity. (Abstr). Proc Am Soc Clin Oncol 6:A340, 1987
Kamen BA, Eibl B, Cashmore A, Bertino J: Uptake and efficacy of trimetrexate, a non-classical antifolate in methotrexate-resistant cells in vitro. Biochem Pharmacol 33:1697–1699, 1984
Ohnoshi T, Ohnuma T, Takahashi I, Scanion K, Kamen BA, Holland JF: Establishment of methotrexate resistant human acute lymphoblastic leukemia cells in culture and effects of folate antagonists. Cancer Res 42:1655–1560, 1982
Mini E, Moroson BA, Franco CT, Bertino JR: Cytotoxic effects of folate-antagonists against methotrexate-resistant human leukemia lymphoblast CCRF-CEM cell line. Cancer Res 45:325–330, 1985
Broome MG, Johnson RK, Wodinsky I: Biochemical and biological characterization of two new antifols, NSC 127755 and TMQ in comparison with methotrexate. (Abstr). Proc AACR 21:309, 1980
NCI Clinical Brochure: Trimetrexate glucoronate NSC 352122, November, 1983
Warner Lambert: Investigator's Brochure. Trimetrexate glucuronate, 1985
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alberts, A.S., Falkson, G., Badat, M. et al. Trimetrexate in advanced carcinoma of the esophagus. Invest New Drugs 6, 319–321 (1988). https://doi.org/10.1007/BF00173651
Issue Date:
DOI: https://doi.org/10.1007/BF00173651