Summary
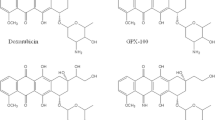
The phase I study of a new anthracycline, 4′-o-tetrahydropyranyl adriamycin, was performed. A dose limiting factor was leukopenia while thrombocytopenia was less frequent and a maximum tolerated dose was determined as 54 mg/m2. Mild gastrointestinal toxicities including anorexia, nausea and vomiting occurred in about half of the patients, while very minimal alopecia was seen in only one patient. A recommended dose for phase II study was established: 40 mg/m2 at 3-week intervals.
Similar content being viewed by others
References
Umezawa H, Takahashi Y, Kinoshita M, Naganawa H, Masuda T, Ishizuka M, Tatsuta K, Takeuchi T: Tetrahydrophyranyl derivatives of daunomycin and adriamycin. J Antibiot 42:1082–1084, 1979
Tsuruo T: Antitumor activity of a tetrahydophyranyl derivative of adriamycin (in Japanese). Can to Kagakuryoho 8:179–180, 1981
Tsuruo T, Iida H, Tsukagoshi S, Sakurai Y: 4′-o-Tetrahydropyranyl adriamycin as a potential new antitumor agent. Cancer Res 42:1462–1467, 1982
Dantchev D, Paintrand M, Hayat M, Bourut C, Mathé G: Low heart and skin toxicity of a tetrahydrophyranyl derivative of adriamycin (THP-ADM) as observed by electron and light microscopy. J Antibiot 32:1085–1086, 1979
Blum RH, Carter SK: Adriamycin, a new anticancer drug with significant clinical activity. Ann Int Med 80:249–259, 1974
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ogawa, M., Miyamoto, H., Inigaki, J. et al. Phase I clinical trial of a new anthracycline: 4′-o-tetrahydropyranyl adriamycin. Invest New Drugs 1, 169–172 (1983). https://doi.org/10.1007/BF00172076
Issue Date:
DOI: https://doi.org/10.1007/BF00172076