Summary
The effect of excitatory amino acids and their antagonists on adenosine and inosine release has been investigated on unstimulated and electrically stimulated hippocampal slices.
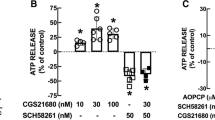
On unstimulated slices N-methyl-D-aspartate (NMDA), quisqualate and glutamate concentration-dependently evoked the release of adenosine and inosine. The effect of NMDA and quisqualate was antagonized by the NMDA receptor antagonist D(−)-2-amino-7-phosphonoheptanoic acid (D-AP7; 100 μmol/1) and the non-NMDA receptor antagonist 6,7-dinitroquinoxaline-2,3-dione (DNQX; 10 μmol/1) respectively. Glutamate (2 and 10 mmol/1)-evoked adenosine and inosine release was not antagonized by the NMDA and non-NMDA receptor antagonists indicating that the effect of glutamate is due to a metabolic rather than a receptor-mediated effect.
Electrical field stimulation at 10 Hz also evoked a release of endogenous adenosine and inosine. Tetrodotoxin (0.5 gmol/1) abolished and absence of Ca2+ markedly reduced the electrically evoked release of adenosine and inosine. Adenosine and inosine release evoked by electrical stimulation at 20 Hz was significantly reduced in the presence of the NMDA receptor antagonist D-AP7, while at 10 Hz no consistent decrease was seen. In the presence of D-AP7 plus DNQX the 10 Hz-evoked adenosine and inosine release was reduced to about half. These data suggest that the electrically evoked release of adenosine and inosine is partly mediated by the release of excitatory amino acids which act at both non-NMDA and NMDA receptors.
Similar content being viewed by others
References
Braas KM, Newby AC, Wilson VS, Snyder SH (1986) Adenosine containing neurons in the brain localized by immunocytochemistry. J Neurosci 6:1952–1961
Burke SP, Nadler JV (1988) Regulation of glutamate and aspartate release from slices of the hippocampal CA1 area: effects of adenosine and baclofen. J Neurochem 51:1541–1551
Corradetti R, Lo Conte G, Moroni F, Passani MB, Pepeu G (1983a) Modulation of the release of excitatory amino acids by adenosine derivatives in the hippocampus. Acta Pharmacol Toxicol 53 [Suppl 1]:111
Corradetti R, Moneti G, Moroni F, Pepeu G, Wieraszko A (1983b) Electrical stimulation of the stratum radiatum increase the release and neosynthesis of aspartate, glutamate, and γ-amino-butyric acid in rat hippocampal slices. J Neurochem 41:1518–1525
Corradetti R, Lo Conte G, Moroni F, Passani MB, Pepeu G (1984) Adenosine decreases aspartate and glutamate release from rat hippocampal slices. Eur J Pharmacol 104:19–26
Cotman CW, Monaghan DT, Ottersen OP, Storm-Mathisen J (1987) Anatomical organization of excitatory amino acid receptors and their pathways. Trends Neurosci 10:273–280
Deckert J, Bisserbe JC, Klein E, Marangos PJ (1988) Adenosine uptake sites in brain: regional distribution of putative subtypes in relationship to adenosine A1 receptors. J Neurosci 8:2338–2349
Fastbom J, Fredholm BB (1985) Inhibition of [3 H] glutamate release from rat hippocampal slices by L-phenylisopropyladenosine. Acta Physiol Scand 125:121–123
Fredholm BB, Sollevi A (1981) The release of adenosine and inosine from canine subcutaneous adipose tissue by nerve stimulation and noradrenaline. J Physiol (Lond) 313:351–367
Goodman RR, Snyder SH (1982) Autoradiographic localization of adenosine receptors in rat brain using [3H] cyclohexiladenosine. J Neurosci 2:1230–1241
Hoehn K, White TD (1990a) Role of excitatory amino acids receptors in K+- and glutamate-evoked release of endogenous adenosine from rat cortical slices. J Neurochem 54:256–265
Hoehn K, White TD (1990b) Glutamate-evoked release of endogenous adenosine from rat cortical synaptosomes is mediated by glutamate uptake and not by receptors. J Neurochem 54:1716–1724
Hoehn K, White TD (1990c) N-methyl-D-aspartate, kainate and quisqualate release endogenous adenosine from rat cortical slices. Neuroscience 39:441–450
Hoehn K, Craig CG, White TD (1990) A comparison of N-methyl-d-aspartate-evoked release of adenosine and [3H] norepinephrine from rat cortical slices. J Pharmacol Exp Ther 255:174–181
Jhamandas H, Dumbrille A (1980) Regional release of 3H adenosine derivatives from rat brain “in vivo”: effect of excitatory amino acids, opiate agonists, and benzodiazepines. Can J Physiol Pharmacol 58:1262–1278
Jones SM, Snell LD, Johnson KM (1987) Phencyclidine selectively inhibits N-methyl-d-aspartate-induced hippocampal [3H] norepinephrine release. J Pharmacol Exp Ther 240:492–497
Jonzon B, Fredholm BB (1985) Release of purines, noradrenaline, and GABA from rat hippocampal slices by field stimulation. J Neurochem 44:217–244
Monaghan DT, Bridges RJ, Cotman CW (1989) The excitatory amino acid receptors. Ann Rev Pharmacol Toxicol 29:365–402
Nowak L, Bregestovski P, Ascher P, Herbet AK, Prochiantz A (1984) Magnesium gates glutamate-activated channels in mouse central neurons. Nature 307:462–464
Padua R, Geiger JD, Dambock S, Nagy JI (1990) 2′-Deoxycoformycin inhibition of adenosine deaminase in rat brain: in vivo and in vitro analysis of specificity, potency, and enzyme recovery. J Neurochem 54:1169–1178
Pedata F, Pazzagli M, Tilli S, Pepeu G (1990) Regional differences in the electrically stimulated release of endogenous and radioactive adenosine and purine derivatives from rat brain slices. NaunynSchmiedeberg's Arch Pharmacol 342:447–453
Perez MTR, Ehinger B (1989) Multiple neurotransmitter systems influence the release of adenosine derivatives from the rabbit retina. Neurochem Int 15:411–420
Perkins MN, Stone TW (1983) “In vivo” release of [3H] purines by quinolinic acid and related compounds. Br J Pharmacol 80:263 -267
Potashner SJ (1978) Effect of tetrodotoxin, calcium and magnesium on the release of amino acids from slices of guinea-pig cerebral cortex. J Neurochem 31:187–195
Prestwich SA, Forda SR, Dolphin AC (1987) Adenosine antagonists increase spontaneous and evoked transmitter release from neuronal cells in culture. Brain Res 405:130–139
Pull I, McIlwain H (1975) Actions of neurohumoral agents and cerebral metabolites on output of adenine derivatives from superfused tissues of the brain. J Neurochem 24:695–700
Watkins JC, Krogsgaard-Larsen P, Honoré T (1990) Structure-activity relationships in the development of excitatory amino acids receptor agonists and competitive antagonists. Trends Pharmacol Sci 11:25–33
Wieraszko A, Lynch G (1979) Stimulation-dependent release of possible transmitter substances from hippocampal slices studied with localized perfusion. Brain Res 160:372–376
Author information
Authors and Affiliations
Additional information
Send offprint requests to F. Pedata at the above address
Rights and permissions
About this article
Cite this article
Pedata, F., Pazzagli, M. & Pepeu, G. Endogenous adenosine release from hippocampal slices excitatory amino acid agonists stimulate release, antagonists reduce the electrically-evoked release. Naunyn-Schmiedeberg's Arch Pharmacol 344, 538–543 (1991). https://doi.org/10.1007/BF00170649
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170649