Summary
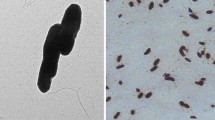
From the anoxic zone of an oil shale leachate column three pyridine-degrading bacterial strains were isolated. Two strains were Gram-negative facultative anaerobic rods and one strain was a branched Gram-positive bacterium. The branched Gram-positive strain had the best pyridine-degrading ability. This organism was aerobic, non-motile, catalase positive, oxidase negative, and had no flagellum. The G+C content of the DNA was 66.5 mol%. The major menaquinone was MK-8(H2). The main cellular fatty acids were saturated and monounsaturated straight chains. This organism contained mycolic acid, meso-diaminopimelic acid, arabinogalactan and glycolyl residues in the cell wall. Due to morphological, physiological and chemotaxonomic characteristics this strain was placed in the genus Rhodococcus. The optimum culture conditions were as follows: temperature 32° C, pH 8.0 and 0.1% v/v of pyridine as sole carbon, energy and nitrogen source. Utilization of pyridine by a batch fermentor culture of Rhodococcus sp. was characterized by a specific growth rate of 0.13 h−1, growth yield of 0.61 mg cell·mg pyridine−1 and a doubling time of 5.3 h−1.
Similar content being viewed by others
References
Becker B, Lechevalier MP, Gordon RE, Lechevalier HA (1964) Rapid differentiation between Nocardia and Streptomyces by paper chromatography of whole-cell hydrolysates. Appl Microbiol 12:421–423
Hugh R, Leifson E (1953) The aromatic significance of fermentative versus oxidative metabolism of carbohydrates by various Gram-negative bacteria. J Bacteriol 66:24–26
Jori A, Calamari D, Cattabeni E, Domenico AD, Galli CL, Galli E, Silano V (1983) Ecotoxicological profile of pyridine. Ecotoxicol Environ Safety 7:251–275
Kroppenstedt RM (1985) Fatty acid and menaquinone analysis of actinomycetes and related organism. Soc Appl Bacteriol Tech Ser 20:173–199
Leenheer JA, Stuber HA (1981) Migration through soil of organic solutes in an oil-shale process water. Environ Sci Technol 15:1467–1475
Mannikin DE, Hutchinson IG, Caldicott AB, Goodfellow M (1980) Thin layer chromatography of methanolysates of mycolic acid-containing bacteria. J Chromatogr 188:221–233
Shukla OP, Kaul SM (1974) A constitutive pyridine degrading system in Corynebacterium sp. Indian J Biochem Biophysics 11:201–207
Shukla OP, Kaul SM (1975) Succinate semialdehyde, an intermediate in the degradation of pyridine by Brevibacterium sp. Indian J Biochem Biophysics 12:326–330
Sims GK, Sommers LE, Konopka A (1986) Degradation of pyridine by Micrococcus leteus isolated from soil. Appl Environ Microbiol 51:963–968
Skerman VBD (1967) A guide to the identification of the genera of bacteria, 2nd edn. Williams and Wilkins, Baltimore
Staneck JL, Roberts GD (1974) Simplified approach to identification of aerobic actinomycetes by thin-layer chromatography. Appl Microbiol 28:226–231
Stuermer DH, Ng DJ, Morris CJ (1982) Organic contaminants in groundwater near an underground coal gasification site in northeastern Wyoming. Environ Sci Technol 16:582–587
Suzuki KI, Komagata K (1983) Taxonomic significance of cellular fatty acid composition in some coryneform bacteria. Int J Syst Bacteriol 33:188–200
Tamaoka J, Komagata K (1984) Determination of DNA base composition by reversed-phase high-performance liquid chromatography. FEMS Microbiol Lett 25:125–128
Uchida K, Aida K (1979) Taxonomic significance of cell-wall acyl type in Corynebacterium-Mycobacterium-Nocarcia group by a glycolate test. J Gen Appl Microbiol 25:169–183
Watson GK, Cain RB (1975) Microbial metabolism of the pyridine ring. Biochem J 146:157–172
Author information
Authors and Affiliations
Additional information
Offprint requests to: S.-T. Lee
Rights and permissions
About this article
Cite this article
Lee, ST., Lee, SB. & Park, YH. Characterization of a pyridine-degrading branched Gram-positive bacterium isolated from the anoxic zone of an oil shale column. Appl Microbiol Biotechnol 35, 824–829 (1991). https://doi.org/10.1007/BF00169903
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00169903