Summary
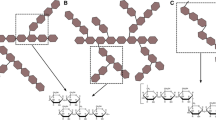
Activity of amylopullulanase from Thermoanaerobacter ethanolicus 39E on α-1,6 and α-1,4-glucosidic linkages in highly branched mammalian glycogen was analyzed by paper chromatography and 13C nuclear magnetic resonance (NMR) spectroscopy. Paper chromatography analysis showed that the glycogen hydrolysate consisted of glucose, maltose, maltotriose and maltotetraose. NMR spectroscopy confirmed that no hydrolysate products of α-1,6 linkage were present resulting from treatment with the amylopullulanase. Therefore, the amylopullulanase efficiently hydrolyzed glycogen both at α-1,6- and at α-1,4-glucosidic linkages into oligosaccharides.
Similar content being viewed by others
References
Coleman, R.D., Yang, S.-S., and McAlister, M.P. (1987). J. Bacteriol. 169, 4302–4307.
Gunja-Smith, Z., Marshall, J.J., Mercier, C., Smith, E.E., and Whelan, W.J. (1970). FEBS Lett. 12, 101–104.
Hyun, H. H., and Zeikus J.G. (1985). Appl.Environ.Microbiol. 49, 1168–1173.
Imanaka, T., and Kuriki, T. (1989). J. Bacteriol. 171, 369–374.
Mathupala, S.P., Saha, B.C., and Zeikus J.G. (1990). Biochem. Biophys. Res. Comm. 166, 126–132.
Mathupala, S.P. (1992). Ph. D. thesis. Michigan State University, U.S.A.
Melasniemi, H. (1987). Biochem.J. 246, 193–197.
Nunez, H.A., Walker, T.E., Fuentes, R., O'Connor, J., Serianni, A., and Barker, R. (1977). J. Supramol. Struct. 6, 535–550.
Plant, A.R., Clemens, R.M., Morgan, H.W., and Daniel, R.M. (1987). Biochem. J. 246, 537–541.
Saha, B.C., Shen, G.-J., Srivastava, K.C., LeCureux, L.W., and Zeikus, J.G. (1989). Enzyme Microb. Technol. 11, 760–764.
Saha, B.C., Lamed, R., Lee, C.-Y., Mathupala, S.P., and Zeikus, J.G. (1990). Appl. Environ. Microbiol. 56, 881–886.
Sata, H., Umeda, H., Kim, C.-H., Taniguchi, H., and Maruyama, Y. (1989). Biochim. Biophys. Acta. 991, 388–394.
Trevelyan, W.E., Procter, D.P., and Harrison J.G. (1950). Nature (London) 166, 444–445.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mathupala, S.P., Park, JH. & Zeikus, J.G. Evidence for α-1,6 and α-1,4-glucosidic bond cleavage in highly branched glycogen by amylopullulanase from Thermoanaerobacter ethanolicus . Biotechnol Lett 16, 1311–1316 (1994). https://doi.org/10.1007/BF00149638
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00149638