Summary
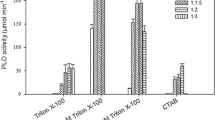
A Fusarium solani pisi recombinant cutinase solubilized in phosphatidylcholine/isooctane reversed micelles was used to catalyse the esterification reaction of butyric acid with 2-butanol at pH 10.7. The influence of temperature, Wo and substrates on lipase stability was evaluated. The enzyme displays a better stability, with a half-life over 125 days, at a temperature of 22°C and for a low water content (WO= 6.5). Butyric acid increased the cutinase deactivation (t1/2=0.56h), while 2-butanol led to a similar half-life (t1/2=14h) as without substrate.
Similar content being viewed by others
References
Henley J.P. and A. Sadana (1985). Enzyme Microb. Technol. 7, 50–60.
Lowry O.H. and Tinsley J.C.(1976). J. Am Oil Chem. Soc. 53, 470–472.
Melo, E.P., Costa S.M.B. and Cabral J.M.S. (1996). Photochem. Photobiol. 63, 000–000 (in press)
Prazeres D.M.F.,Garcia F.A.P. and Cabral J.M.S. (1992). J. Chem. Tech. Biotechnol. 53, 159–164.
Sebastião M.J., Cabral J.M.S. and Aires-Barros M.R. (1993). Biotechnol. Bioeng. 42, 326–332.
Shimada Y., Tominaga Y., Iwai M. and Tsujisak Y. (1983). J. Biochem. 93, 1655–1660.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pinto-Sousa, A.M., Cabral, J.M.S. & Aires-Barros, M.R. Stability of a Fusarium solani pisi recombinant cutinase in phosphatidylcholine reversed micelles. Biotechnol Lett 18, 583–586 (1996). https://doi.org/10.1007/BF00140207
Issue Date:
DOI: https://doi.org/10.1007/BF00140207