Abstract
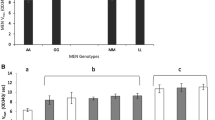
An intraspecific comparison of α-glycerophosphate (α-GPDH: E.C.1.1.1.8) and alcohol dehydrogenase (ADH: E.C.1.1.1.1) enzyme activity levels was carried out in Drosophila melanogaster. The results indicate that (1) α-GPDH is a relatively conservative and ADH a relatively variable enzyme system with regard to structurally determined activity variation but that (2) the conservative nature of α-GPDH activity variation does not extend to the intra-genotypic level. The results are consistent with the view that different kinds of selective pressures are being exerted on the enzyme's structural and modifier gene loci.
Similar content being viewed by others
References
Avise, J. & J. McDonald, 1976. Enzyme changes during development of holo- and hemi-metabolic insects. Comp. Biochem. Physiol. 53B 393–397.
Ayala, F., J. Powell, M. Tracey, C. Mourao & S. Perez-Scalas, 1972. Enzyme variability in the Drosophila willistoni group. IV. Genic variation in natural populations. Genetics 70: 113–139.
Ayala, F. & J. Powell, 1972. Enzyme Variability in the Drosophila willistoni Group VI. Levels of polymorphism and the physiological function of enzymes. Biochem. Genet. 7: 331–345.
Barnes, B. & A. Birley, 1978. Genetic variation for enzyme activity in a population of Drosophila melanogaster. Heredity 40: 51–57.
Bewley, G., 1978. Heat stability studies at the α-glycerophosphate dehydrogenase locus in Drosophila melanogaster. Biochem. Genet. 16: 769–775.
Gillespie, J. & K. Kojuma, 1968. The degree of polymorphism in enzymes involved in energy production compared to that in nonspecific enzymes in two Drosophila annanassae populations. Proc. natn. Acad. Sci. U.S.A. 61: 583–590.
Hedrick, P. & J. McDonald, 1980. Regulatory gene adaptation: an evolutionary model. Heredity in press.
Laurie-Ahlberg, C., G. Maroni, G. Bewley, J. Lucchesi & B. Weir, 1980. Quantitative genetic variation of enzyme activities in natural populations of Drosophila melanogaster. Proc. natn. Acad. Sci. U.S.A. (in press).
Lakovaara, S., A. Saura, & P. Lankinen, 1977. Evolution at the α-GPDH locus in Drosophilidue. Evolution 31: 319–330.
McDonald, J. & J. Avise, 1976. Evidence for the adaptive significance of enzyme activity levels: Interspecific variation in α-GPDH and ADH in Drosophila. Biochem. Genet. 14: 347–355.
McDonald, J. & F. Ayala, 1978. Genetic and biochemical basis of enzyme activity variation in natural populations I. Alcohol dehydrogenase in Drosophila melanogaster. Genetics 89: 371–388.
Rawls, J. & JK. Lucchesi, 1974. Regulation of enzyme activities in Drosophila I. The detection of regulatory loci by gene dosage responses. Genet. Res., 24: 59–72.
Sacktor, B. & D. Cochran, 1957. DPN-specific α-glycerophosphate dehydrogenase in insect flight muscle. Biochim. Biophys. Acta 25: 699–653.
Sacktor, B., 1965. Energetics and respiratory metabolism of muscular contraction. In: Rockstein, M. (ed.) Physiology of Insecta, vol. 2, Academic Press, N. Y.: 483–580.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wilson, P.G., McDonald, J.F. A comparative study of enzyme activity variation between α-glycerophosphate and alcoholdehydrogenases in Drosophila melanogaster. Genetica 55, 75–79 (1981). https://doi.org/10.1007/BF00134008
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00134008