Abstract
Position effect variegation in Drosophila melanogaster is associated with the inability of certain genes to be correctly expressed in a proportion of cells, giving a mosaic phenotype. The lack of expression is thought to be due to alterations in the gene's chromatin structure due to its proximity to a region of heterochromatin. Because of the difficulties involved, there is little biochemical data to support the intuitively appealing model of ‘heterochromatin spreading’ used to explain this phenomenon.
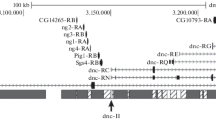
Differences in restriction fragment length were used to distinguish DNA regions from either normal (non-position affected) or rearranged (position affected) chromosomes so as to examine possible changes in gene copy number and the effects of endogenous nucleases. DNA sequences at the breakpoint of In (1)w m4, which variegates for the white gene, were assayed under conditions where the chromatin conformation was altered using second site modifier mutations (Su(var) or En(var)). No change in the DNA sequerice copy number was observed at either chromosome breakpoint, relative to wild type, when either suppressor or enhancer mutations were present. Therefore copy number change, through differential polyploidization or somatic gene loss, is not affected by Su(var) or En(var) induced changes in the chromatin conformation.
Initial experiments showed a gross difference in the sensitivity of DNA to endogenous nucleases that appeared associated with Su(var) and En(var) mutations. En(var) mutation bearing samples appeared delayed in the digestion, relative to Su(var). This differential sensitivity seemed to be genome-wide as there was no detectable difference between either breakpoint of In(1)w m4 or the sequences on the homologous w - chromosome. However, after isogenizing the genetic background, the previously noted difference between the Su(var) and En(var) mutations was eliminated. In studies dealing with nuclease digestion of chromatin, the isogenization of genetic background is essential before meaningful comparisons can be made.
Similar content being viewed by others
References
AnanievE. V. & V. A.Gvozdev, 1974. Changed pattern of transcription and replication in polytene chromosome of Drosophila melanogaster resulting from eu-heterochromatin rearrangement. Chromosoma 45: 173–191.
EissenbergJ. C., 1989. Position effect variegation in Drosophila: Towards a genetics of chromatin assembly. Bioessays 11: 12–17.
EissenbergJ. C. & S. C. R.Elgin, 1991. Boundary functions in the control of gene expression. T. I. G. 7: 335–340.
EissenbergJ. C., T. C.James, D. M.Foster-Hartnett, T.Hartnett, V.Ngan & C. R.Elgin, 1990. Mutation in a heterochromatin-specific chromosomal protein is associated with suppression of position-effect variegation in Drosophila melanogaster. Proc. Natl. Acad. Sci USA 87: 9923–9927.
GeyerP. K. & V. G.Corces, 1987. Separate regulatory elements are responsible for the complex pattern of tissue-specific and developmental transcription of the yellow locus in Drosophila melanogaster. Genes & Dev. 1: 996–1004.
GrigliattiT., 1991. Position-effect variegation — an assay for nonhistone chromosomal proteins and chromatin assembly and modifying factors. Functional Organization of the Nucleus: A laboratory guide. Academic Press, Inc., San Diego.
Hartmann-GoldsteinI. J., 1967. On the relationship between heterochromatinization and variegation in Drosophila with special reference to temperature sensitive periods. Genet. Res. 10: 143–159.
HayashiS., A.Ruddell, D.Sinclair & T.Grigliatti, 1990. Chromosomal structure is altered by mutations that suppress or enhance position effect variegation. Chromosoma 99: 391–400.
HenikoffS., 1981. Position effect variegation and chromosome structure of a heat shock puff in Drosophila. Chromosoma 83: 381–393.
KarpenG. H. & A. C.Spradling, 1990. Reduced DNA polytenization of a mini chromosome region undergoing position-effect variegation in Drosophila. Cell 63: 97–107.
KellumR. & P.Schedl, 1991. A position-effect assay for boundaries of higher order chromosomal domains. Cell 64: 941–950.
LefevreG. & M. M.Green, 1972. Genetic duplication in the white-split interval of the X chromosome in Drosophila melanogaster. Chromosoma (Berl.) 36: 391–412.
LindsleyD. L. & E. H.Grell, 1968. Genetic Variations of Drosophila melanogaster. Carnegie Institution of Washington, Oak Ridge.
LindsleyD. L. & G. G.Zimm, 1992. The genome of Drosophila melanogaster. Academic Press, Inc., San Diego.
LockeI., M. A.Kotarski & K. D.Tartof, 1988. Dosage-dependent modifiers of position effect variegation in Drosophila and a mass action model that explains their effect. Genetics 120: 181–108.
Locke, J., S. Hanna & D. Kong, 1003. Evidence for mobilization of hobo transposons in a P-element mutagenesis screen. Genome. (In Press).
ManiatisT., E. F.Frisch & J.Sambrook, 1982. Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor.
ReuterG., R.Dorn, G.Wustmann & G.Bauh, 1986. Third chromosome suppressor of position-effect variegation loci in Drosophila melanogaster. Mol. Gen. Genet. 202: 481–487.
RubinG. M. & A. C.Spradling, 1989. Vectors for P-element-mediated gene transfer in Drosophila melanogaster. Nucleic Acids Res. 11: 6341–6351.
SambrookJ., E. F.Fritsch & T.Maniatis, 1989. Molecular Cloning: A laboratory Manual, 2nd Edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York.
SpoffordJ. B., 1976. Position effect variegation in Drosophila. Genetics and Biology of Drosophila. Vol. 1C. Academic Press, London.
SpradlingA. C. & G. H.Karpen, 1990. Sixty years of mystery. Genetics 126: 770–784.
TartofK. D., C.Bishop, M.Jones, C. A.Hobbs & J.Locke, 1989. Towards and understanding of position effect variegation. Dev. Genetics 10: 162–176.
TartofK. D., C.Hobbs & M.Jones, 1984. A structural basis for variegating position effects. Cell 37: 860–878.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Locke, J. Examination of DNA sequences undergoing chromatin conformation changes at a variegating breakpoint in Drosophila melanogaster . Genetica 92, 33–41 (1993). https://doi.org/10.1007/BF00057505
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00057505