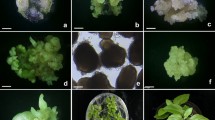
Abstract
Protocols were established for achieving plant regeneration from stem internode, callus, and cell suspension cultures of Solanum lycopersicoides Dun. Two accessions of S. lycopersicoides exhibited different responses as to callus formation on various media, requirement of gibberellic acid for shoot regeneration, and ability to grow in suspension culture. The optimum medium for initiation and maintenance of cell suspension cultures was Murashige and Skoog [9] medium with 15 mg l− NAA. For shoot regeneration, of three cytokinins tested, zeatin was found most effective relative to number, rapidity of response and overall quality of shoots. Shoot regeneration from stem explants, callus and suspension cultures was optimum on MS + 3.0 mg l−1 zeatin + 0.1 mg l−1 gibberellic acid.
Similar content being viewed by others

References
Breidenbach RW, Waring AJ (1977) Response to chilling of tomato seedlings and cells in suspension cultures. Plant Physiol 60:190–192
Gleddie S, Keller W, Setterfield G (1983) Somatic embryogenesis and plant regeneration from leaf explants and cell suspensions of Solanum melongena (eggplant). Can J Bot 61:656–666
Halperin W, Wetherell F (1964) Adventive embryony in tissue cultures of the wild carrot, Daucus carota. Am J Bot 51:274–283
Herner RC (1982) Chilling injury in tomato. Report to a symposium: Plant responses to environmental stress. Annual meeting AAAS. Washington
Herner RC, Kamps T (1983) Chilling injury tolerance of wild tomato species. Proc. 4th Tomato Quality Workshop. March 7–10. Veg Crops Res RPT VEC-83-1
Mariotti D, Arcioni S (1983) Callus culture of Coronilla varia L. (crownvetch): Plant regeneration through somatic embryogenesis. Plant Cell Tissue Organ Culture 2:103–110
Matsuoka H, Kokichi H (1979) NAA-induced organogenesis and embryogenesis in hypocotyl callus of Solanum melongena L. J Exp Bot 30:363–370
Meredith CP (1979) Shoot development in established callus cultures of cultivated tomato (Lycopersicon esculentum Mill.). Z. Pflanzenphysiol 95:405–411
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Patterson BD, Paull R and Smillie RM (1978) Chilling resistance in Lycopersicon hirsutum Humb. & Bonpl., a wild tomato with a wide altitudinal distribution. Aust J Plant Physiol 5:609–617
Rick CM (1951) Hybrids between Lycopersicon esculentum Mill. and Solanum lycopersicoides Dun. Proc Nat Acad Sci 37:741–744
Santos AVP Dos, Outka DE, Cocking EC, Davey MR (1980) Organogenesis and somatic embryogenesis in tissues derived from leaf protoplasts and leaf explants of Medicago sativa. Z Pflanzenphysiol 99:261–270
Scott SJ, Jones RA (1982) Low temperature seed germination of Lycopersicon species evaluated by survival analysis. Euphytica 31:869–883
Sung ZR, Smith R, Horowitz J (1979) Quantitative studies of embryogenesis in normal and 5-methyltryptophan-resistant cell lines of wild carrot. The effects of growth regulators. Planta 147:236–240
Uchimiya H, Murashige T (1974) Evaluation of parameters in the isolation of viable protoplasts from cultured tobacco cells. Plant Physiol 54:936–944
Vallejos CE (1979) Genetic diversity of plants for response to low temperature and its potential use of crop plants. In: Lyons JM, Graham D, Raison JK (eds) Low Temperature Stress in Crop Plants, the Role of the Membrane. Academic Press, New York, pp. 473–489
Author information
Authors and Affiliations
Additional information
Michigan Agricultural Experiment Station Journal Article No. 11589.
Rights and permissions
About this article
Cite this article
Handley, L.W., Sink, K.C. Plant regeneration of Solanum lycopersicoides Dun. from stem explants, callus and suspension cultures. Plant Cell Tiss Organ Cult 5, 129–138 (1985). https://doi.org/10.1007/BF00040309
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00040309